Home

March 25, 2026
Bad Homburg, Germany
Publication Annual Report 2025
Consolidated Financial Statements and Management Report (IFRS)
With our reporting and communication formats, we provide comprehensive information on our Sustainability Framework and the management of material topics. Group-wide policies enable consistent integration of sustainability considerations in daily business operations.
In our rating overview, we present the latest scores from relevant ESG rating agencies.
In our annual sustainability reporting, we disclose required regulatory information regarding our material sustainability topics. In addition, we publish our Sustainability Highlights Magazine once a year, providing an overview of our related activities.
Sustainability Statement 2025
in accordance with the European Sustainability Reporting Standards
Our policies provide employees and business partners with clear guidance on our approach to responsible business conduct and sustainability. Among the relevant internal documents are:
Fresenius Group Code of Coduct
ESG rating agencies regularly evaluate and review Fresenius' sustainability performance. In doing so, they focus on different aspects across the areas of Environment, Social, and Governance (ESG).
A close dialog with our stakeholders on these topics is very important to us. We are actively working to enhance transparency on relevant topics. This is reflected in consistently good rating results.
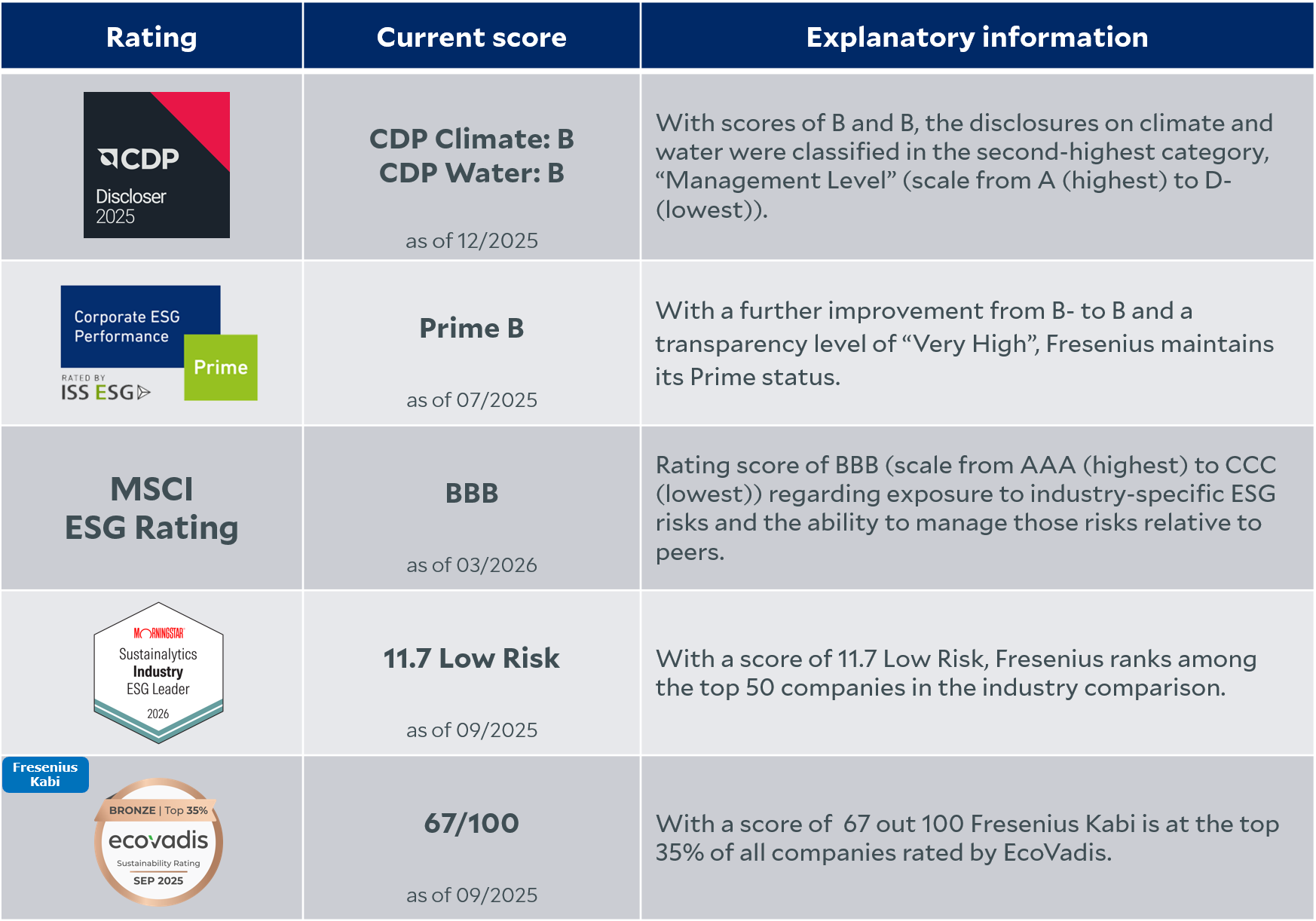
*The use by FRESENIUS SE & CO. KGAA of any MSCI ESG RESEARCH LLC or its affiliates (“MSCI”) data, and the use of MSCI logos, trademarks, service marks or index names herein, do not constitute a sponsorship, endorsement, recommendation, or promotion of FRESENIUS SE & CO. KGAA by MSCI. MSCI services and data are the property of MSCI or its information providers, and are provided ‘as-is’ and without warranty. MSCI names and logos are trademarks or service marks of MSCI.
*Copyright ©2025 Morningstar Sustainalytics. All rights reserved. The information, data, analyses and opinions contained herein: (1) includes the proprietary information of Sustainalytics and/or its content providers; (2) may not be copied or redistributed except as specifically authorized; (3) do not constitute investment advice nor an endorsement of any product, project, investment strategy or consideration of any particular environmental, social or governance related issues as part of any investment strategy; (4) are provided solely for informational purposes; and (5) are not warranted to be complete, accurate or timely. The ESG-related information, methodologies, tool, ratings, data, and opinions contained or reflected herein are not directed to or intended for use or distribution to India-based clients or users and their distribution to Indian resident individuals or entities is not permitted. Neither Morningstar Inc., Sustainalytics, nor their content providers accept any liability for the use of the information, for actions of third parties in respect to the information, nor are responsible for any trading decisions, damages or other losses related to the information or its use. The use of the data is subject to conditions available at https://www.sustainalytics.com/legal-disclaimers.
Our commitment to a healthy planet
As a healthcare company, we operate in a special field of tension. On the one hand, we want to do our best to reduce potentially adverse environmental impacts to a minimum. On the other hand, we must never lose sight of the strict requirements that are placed on patients’ safety and hygiene. Our aim is to promote human health while further reducing our ecological footprint.
People need a healthy home – today and tomorrow. By reducing the environmental impact of our activities, we want to play our part in mitigating climate change and conserving natural resources.

We have a clear goal: to decarbonize our operations and our value chain.
Fresenius manufactures medical products and operates healthcare facilities, which inevitably results in energy consumption and associated greenhouse gas emissions. In production, for example, the machines and containers have to be sterilized, and in our hospitals a wide variety of technical systems are in constant operation. This presents us with special challenges: On the one hand, we want to reduce our energy demand as far as possible and on the other hand, we must always guarantee the safety of patients in our facilities and ensure a stable supply of energy in our production.
We want to live up to our responsibilities and help achieve the goal of the Paris Climate Agreement: Our climate targets aim to limit global warming to 1.5 °C. All our climate protection activities contribute to our long-term objective of attaining net zero by 2050: This requires a company to reduce its avoidable greenhouse gas emissions, while unavoidable emissions must be offset by removing an equivalent amount from the atmosphere and storing it for the long term.
Within our own operations, we are committed to reducing our Scope 1 and Scope 2 emissions by 50% by 2030.
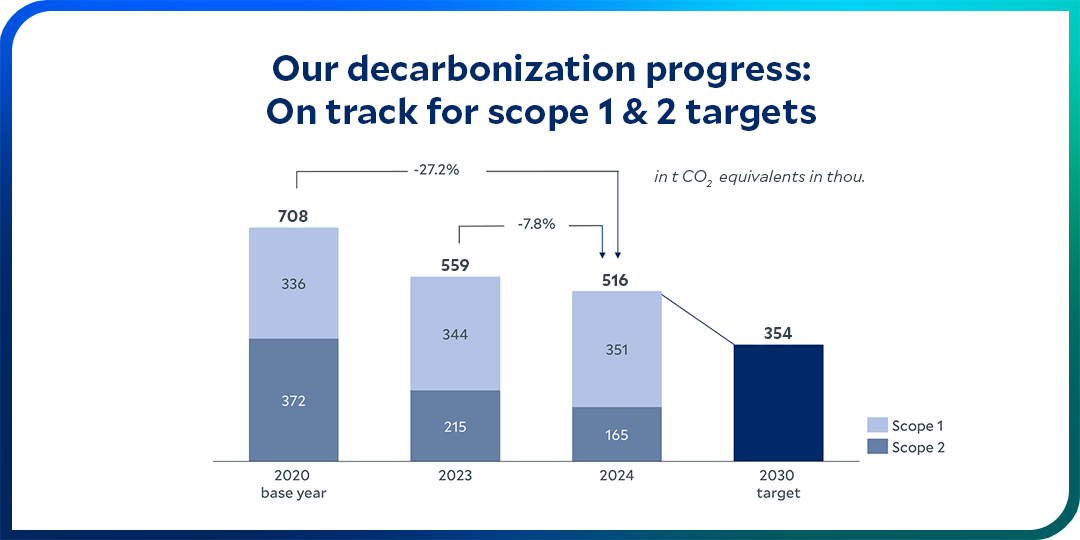
The decarbonization of our value chain is a decisive factor on our long-term path to net zero. Scope 3 emissions include greenhouse gases that are released indirectly in our upstream and downstream value chains – such as from purchased raw materials or at the end of the life of sold products.
Climate change is also a health issue. Rising temperatures, more frequent heatwaves and changing weather patterns directly affect people's health, particularly vulnerable groups such as the elderly, chronically ill, and children. Heat stress can lead to circulatory problems, dehydration and an increased risk of death. At the same time, climate change contributes to the spread of infectious diseases, for example by introducing new vectors such as mosquitoes or contaminating water sources. As a healthcare company, we therefore consider it our responsibility to not only react to the effects of climate change, but also actively contribute to mitigating it. After all, climate protection is health protection.

We place particular emphasis on preserving water quality. In water-stressed areas, we are especially committed to reducing our water withdrawal.

Water is one of our most valuable resources. As a healthcare company, we are dependent on water: We need drinking water of the highest quality to ensure safe patient care. It is crucial for hygiene and well-being. We also use water in the production of our pharmaceutical products – as process water as well as product component. The quality requirements for this water are even higher than those for drinking water.

It is our ambition to avoid or minimize any negative impact on the environment that may arise from our direct business operations or from downstream activities. This also includes avoiding unnecessarily polluting the sources from which we obtain water or into which we discharge our wastewater.
At Fresenius, we focus on reducing our waste and embedding circular principles into product design and packaging solutions.
We are committed to conserving natural resources wherever possible. In doing so, we always have to strike a balance between resource efficiency and hygiene requirements. Disposable items are often used in clinics for hygienic reasons. Our options for saving resources are limited here. There are also strict regulations for pharmaceutical packaging. Our most important levers for conserving resources are therefore developing durable and resource-saving products, reusing resources wherever possible, and disposing of waste safely and systematically.


Explore our stories to learn what sustainability looks like in our daily operations:
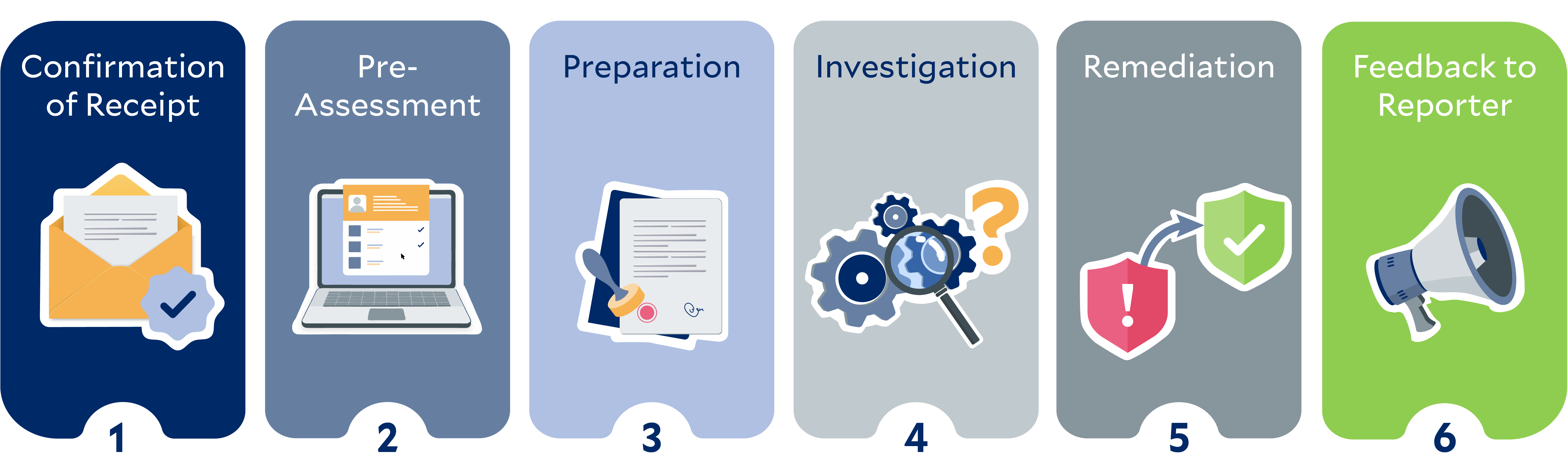
We value open communication and strive to create an environment in which patients, employees, members of local communities, business partners, or other potentially affected persons can report human rights violations or non-compliance with environmental regulations. To this end, we have set up whistleblower systems.
The following graphic illustrates the steps for processing reports, starting from the acknowledgment of receipt to the feedback provided to the person filing the report, using the example of two fictional protagonists: Kate and Aditi.*
*The persons and stories depicted in this image are purely fictional. Any resemblance to actual persons is purely coincidental and unintentional.
Additional information on our grievance mechanisms and the protection of whistleblowers can be found in the process descriptions of the respective companies:
Fresenius SE:
https://www.fresenius.com/grievance-mechanism
Fresenius Kabi:
https://www.fresenius-kabi.com/responsibility/business-ethics
Fresenius Helios:
https://www.helios-gesundheit.de/menschenrechte
Quirónsalud:
https://www.quironsalud.com/en/group/whistleblowing-channel
Have you ever had the feeling that something isn’t quite right? Perhaps you noticed something that shouldn’t be the way it is, or you yourself were affected by such a situation. That’s when it’s time to report a grievance.
Watch the video to find out how to report a grievance, to learn why reporting grievances is so important, and to see what happens to a report after you submit it.
The metrics describe the reports received through our reporting systems in the reporting year that were related to human rights – broken down into those affected in our own operations and those in our value chain. Of the 24 (2024: 28) reports received, 1 (2024: 4) proved to be a human rights violation. No report was related to a severe human rights incident in the upstream or downstream value chain or in Fresenius’ own operations.1
| Own operations | Value chain | |
|---|---|---|
Reports received with human rights relevance | 23 | 1 |
Of which are violations | 1 | - |
Of which are severe human rights violations | - | - |
1Severe human rights violation include incidents of forced labor, incidents of human trafficking, incidents of child labor as well incidents involving a large number of people or affecting a large area. This categorization is based, for example, on the definitions of the Corporate Sustainability Reporting Directive (CSRD).
The Group-wide Sustainability Report 2025 (CSRD Report) contains detailed information on the substantiated cases and the remedial and preventive measures implemented in the chapters “Own workforce” and “Workers in the value chain”.
At Fresenius, we attach great importance to taking appropriate account of the interests of whistleblowers and those of potentially affected persons in the processing of reports. We achieve this through a transparent, fair and comprehensive examination of all incoming complaints by specially trained staff.
Our aim is to ensure that the concerns of those affected are taken seriously and processed in accordance with the applicable legal and internal company requirements. In doing so, we ensure that all relevant information and perspectives are included in the decision-making process to find a balanced and appropriate solution.
Through feedback from those affected, we analyze how our process is accepted in practice and where there is potential for optimization. This input is incorporated into the further development of our processes to ensure that the interests of those affected are taken into account even better in future.
The results of our risk analysis and the findings on potential target groups of our grievance and whistleblower channels are incorporated into the further development of our grievance mechanism and the processing of respective reports. Based on our findings, we review the effectiveness of the procedure on an annual basis, or more frequently if required. Where necessary, we make appropriate adjustments and changes with regard to accessibility and process.

Human Rights at Fresenius
Our Human Rights ProgramContact
Fresenius SE & Co. KGaA
Else-Kröner-Str. 1
61352 Bad Homburg
Germany
humanrights@fresenius.com
Human Rights Program
Our Human Rights Program Group-wide Governance & Responsibilities Risk Assessment & Impact Preventive & Remedial ActionYou want to file a report?
Reports on possible human rights violations or other types of compliance violations can be reported around the clock, either anonymously or by name, via our whistleblower system*:
Whistleblower system
https://freseniusgroup.ethicspoint.com
By telephone*
+49 (0) 800 181 1338
By mail
Fresenius SE & Co. KGgaA Business Integrity
Else-Kröner-Str. 1
61352 Bad Homburg v.d.H. Deutschland
By e-mail:
humanrights@fresenius.com
* The prices of your mobile or landline contract apply.
The reporting on the respect for human rights for the year 2024 and following can be found in our Sustainability Report (CSRD Report). Further information on Fresenius' human rights program is available here: fresenius.com/en/human-rights
Human rights risks can change over time. We therefore conduct annual and event-related risk analyses. In the 2024 reporting period, this was done in the third and fourth quarters. An event-related risk analysis was not conducted.
Remark: The results of the risk analysis for the 2025 reporting year will be updated at a later stage.
Identifying and assessing human rights risks in our own company and in our value chain is a comprehensive process that consists of risk identification, risk analysis, and risk assessment. We follow a risk-based approach that can be divided into three phases and is explained below with the help of the fictional protagonist, John.*
*The persons and stories depicted in these images are purely fictional. Any resemblance to actual persons is purely coincidental and unintentional.
To weight and prioritize risks, we introduced a comprehensive methodology for assessing their impact and likelihood of occurring. Using this method, the risks are plotted on a matrix (4x4).
The evaluated impact on those affected ranges from “low” to “severe”. It is assessed using four evaluation criteria: scope, scale, possibility for remedial action, and company involvement.
Probability of occurrence ranges from “unlikely” to “almost certain”. It is assessed using three different evaluation criteria: process evaluation, evaluation of similar cases which have already arisen, and context factors which could increase the likelihood of the risks materializing.
We carry out a regular risk analysis of human rights topics for both our own businesses and our supply chains in accordance with the specific requirements of applicable international and national laws and regulations. As part of this risk analysis, we identify areas that we consider to have high priority due to the potential severity of possible violations and due to our ability to influence them.
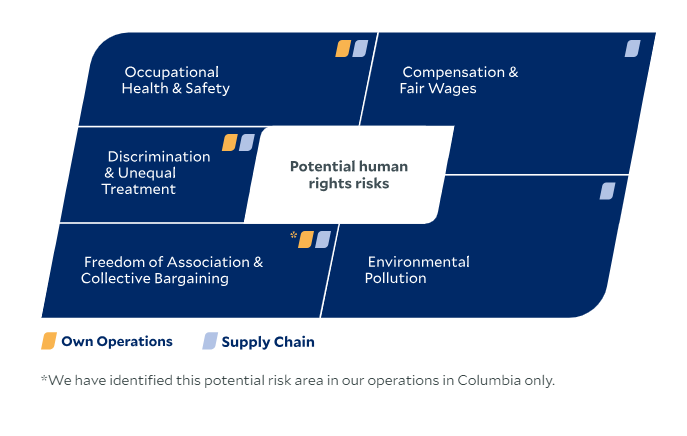
The risk analysis for the 2024 reporting year confirmed the following existing prioritized risk areas for our own operations and revealed additional potential risks:
As a healthcare Group, we not only bear responsibility for the well-being of our patients, but also for the health and safety of our employees. We implemented a Group Policy on Social and Labor Standards. The guideline describes our global social and labor law minimum standards. We expect our employees and managers in all business segments of the Fresenius Group to comply with this guideline without exception. Lower standards are not acceptable. Should laws or practices in countries where we operate restrict or contradict the standards set out in this policy, we will apply the policy to the extent permitted by local laws . Moreover, the Fresenius Code of Conduct stipulates that we take the necessary measures to protect our employees and prevent work-related accidents and illnesses.
Creating a safe and healthy working environment is a priority for us. When it comes to health protection, prevention is our basic principle: We therefore provide our employees with comprehensive programs to promote their health and prevent work-related illnesses. The return of employees after an illness is regulated, for example, by the company integration management system.
We have introduced numerous management systems and measures throughout the Group and adapted them to the business models of the business segments. They focus on occupational health and safety in the production area as well as occupational health management for employees in healthcare facilities or in administration.
All locations are also subject to the respective local regulations and laws. Compliance with these regulations is ensured at local level. In addition to statutory regulations, internal guidelines and directives such as management manuals and standard operating procedures also play a significant role in occupational health and safety. In addition to the Group-wide Fresenius Code of Conduct, the business segments have their own guidelines that regulate occupational health and safety, e.g. the Clinical Code of Conduct for the rehabilitation and nursing units and medical personnel in the healthcare services market segment.
The internal requirements are supplemented by corresponding internationally recognized standards for management systems such as ISO 45001 at some locations as well as other certifications in accordance with ISO or national standards. The overarching aim of the ISO 45001 management system is to continuously improve occupational health and safety management, align it with internationally recognized methods and ensure the effectiveness of existing procedures and systems. To drive this forward, we are consistently expanding the number of entities certified to this standard.
Further Information can be found in the 2024 CSRD report from page 216.
To minimize potential human rights risks connected to our business activities, we have taken further preventive and mitigating measures. These include, for example, training that we offer to our workforce. The Fresenius Group, for instance, conducts regular occupational health and safety training to prevent incidents in its fields of operation.
To sustainably promote tolerance and appreciation within our teams in the long-term and to mitigate the risk of discrimination, it furthermore is not only necessary to have a corresponding culture that is exemplified by the management bodies; employees also receive training and further education on the topic of diversity.
The elimination of discrimination is both a component of our Group-wide compliance programs and a key element of our Human Rights Program. These concepts are supplemented by suitable controls, process documentation, training concepts, awareness-raising measures, and the use of whistleblower systems. In this way, we want to ensure that discrimination, including harassment, is prevented, contained, or combated in our operational business if we become aware of violations, risks, or impacts.
Another central element is the successive roll out of a company-wide human rights training for our employees. The training aims at educating our employees on this important topic – not only about their personal human rights, but also about the contribution that everyone can make in their daily work. It further imparts knowledge about individual rights and how to deal with possible human rights violations.
In addition, we have implemented individual and local measures to provide our employees with the best possible protection within our sphere of influence. For instance, at high-risk locations, we deploy more security staff than is typically found in hospitals to better protect our employees and patients around the clock. Furthermore, a monthly committee with participants from local management and employees has been established to strengthen the dialogue.
Further details on preventive and remedial measures, as well as on appropriateness and effectiveness, are available here.
The risk analysis for the 2024 reporting year confirmed the following existing prioritized risk areas for our value chain and revealed additional potential risks:
To counter the potentially negative effects, we have initiated and implemented further preventive measures in addition to existing ones. With the risk-based implementation of human rights and environmental clauses in contracts, we also agree with suppliers on specific requirements for cooperation and information obligations in the event of human rights violations.
Our Code of Conduct for Business Partners also sets out fundamental expectations regarding respect for human rights. To monitor compliance with these principles and use the results to provide industry-wide support, Fresenius Kabi, for example, prepared to join the Pharmaceutical Supply Chain Initiative (PSCI) as an associate member in the reporting year (2024). In the future, the company will participate in the industry-wide audit pooling and thus contribute to greater transparency regarding working conditions and – where necessary – corresponding corrective or remedial measures in the pharmaceutical supply chain.

Marco Kraemer, Director Supplier Quality Management & Human Rights Function, Fresenius Kabi
To further increase transparency in our upstream and downstream processes, we also plan to further expand the existing descriptions and visualizations of our value chains and carry out focus risk analyses on this basis. Further details on preventive and remedial measures, as well as on appropriateness and effectiveness, are available here.

Human Rights at Fresenius
Our Human Rights ProgramContact
Fresenius SE & Co. KGaA
Else-Kröner-Str. 1
61352 Bad Homburg
Germany
humanrights@fresenius.com
Human Rights Program
Our Human Rights Program Group-wide Governance & Responsibilities Preventive & Remedial Action Grievance Procedure & HandlingWhistleblowing system
Reports on possible human rights or other types of compliance violations can be reported around the clock, either anonymously or by name, via our whistleblower system*:
Phone number: +49 (0) 800 181 1338*
https://freseniusgroup.ethicspoint.com
More infos about our grievance mechanism
*The prices of your mobile or landline contract apply
The reporting on the respect for human rights for the year 2024 and following can be found in our Sustainability Report (CSRD Report). Further information on Fresenius' human rights program is available here: fresenius.com/en/human-rights
S&P Global Ratings (S&P), a globally recognized credit rating agency today revised its credit outlook for Fresenius SE from stable to positive. The rating was affirmed at BBB. As part of its evaluation, S&P acknowledged the significant progress highlighting Fresenius’ strong operating performance, particularly within its growth vectors, ongoing cost base improvements, as well as a further reduction in the Company’s leverage. S&P also highlighted the sharpened and simplified portfolio, underscoring the Company’s resilience in the current operating environment.
“The revised outlook is another proof point that #FutureFresenius is paying off. It confirms our focus on long-term profitable growth and balance sheet strength, while at the same time preparing the business for future growth. Based on the strength of our operating businesses and the strong cash flow generation, we have significantly deleveraged the Company over the past years and expect to stay well within our self-imposed target leverage range,” says Fresenius CFO Sara Hennicken.
Fresenius is rated investment grade by the three leading credit rating agencies S&P Global Ratings (BBB/positive), Moody’s (Baa3/stable) and Fitch (BBB-/stable). The company is committed to its investment grade rating and to its self-imposed target leverage range of 2.5 to 3.0x net debt/EBITDA1, which forms part of its capital allocation framework.
1 At average exchange rates for both net debt and EBITDA; pro forma closed acquisitions/divestitures; before special items; including lease liabilities and Fresenius Medical Care dividend, net debt adjusted for the valuation effect of the exchangeable bond
This release contains forward-looking statements that are subject to various risks and uncertainties. Future results could differ materially from those described in these forward-looking statements due to certain factors, e.g. changes in business, economic and competitive conditions, regulatory reforms, results of clinical trials, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, the availability of financing and unforeseen impacts of international conflicts. Fresenius does not undertake any responsibility to update the forward-looking statements in this release.
Fresenius SE & Co. KGaA Registered Office: Bad Homburg, Germany / Commercial Register: Amtsgericht Bad Homburg, HRB 11852
Chairman of the Supervisory Board: Wolfgang Kirsch
General Partner: Fresenius Management SE
Registered Office: Bad Homburg, Germany / Commercial Register: Amtsgericht Bad Homburg, HRB 11673 Management Board: Michael Sen (Chairman), Pierluigi Antonelli, Sara Hennicken, Robert Möller, Dr. Michael Moser
Chairman of the Supervisory Board: Wolfgang Kirsch
S&P Global Ratings (S&P), a globally recognized credit rating agency today revised its credit outlook for Fresenius SE from stable to positive. The rating was affirmed at BBB. As part of its evaluation, S&P acknowledged the significant progress highlighting Fresenius’ strong operating performance, particularly within its growth vectors, ongoing cost base improvements, as well as a further reduction in the Company’s leverage. S&P also highlighted the sharpened and simplified portfolio, underscoring the Company’s resilience in the current operating environment.
“The revised outlook is another proof point that #FutureFresenius is paying off. It confirms our focus on long-term profitable growth and balance sheet strength, while at the same time preparing the business for future growth. Based on the strength of our operating businesses and the strong cash flow generation, we have significantly deleveraged the Company over the past years and expect to stay well within our self-imposed target leverage range,” says Fresenius CFO Sara Hennicken.
Fresenius is rated investment grade by the three leading credit rating agencies S&P Global Ratings (BBB/positive), Moody’s (Baa3/stable) and Fitch (BBB-/stable). The company is committed to its investment grade rating and to its self-imposed target leverage range of 2.5 to 3.0x net debt/EBITDA1, which forms part of its capital allocation framework.
1 At average exchange rates for both net debt and EBITDA; pro forma closed acquisitions/divestitures; before special items; including lease liabilities and Fresenius Medical Care dividend, net debt adjusted for the valuation effect of the exchangeable bond
This release contains forward-looking statements that are subject to various risks and uncertainties. Future results could differ materially from those described in these forward-looking statements due to certain factors, e.g. changes in business, economic and competitive conditions, regulatory reforms, results of clinical trials, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, the availability of financing and unforeseen impacts of international conflicts. Fresenius does not undertake any responsibility to update the forward-looking statements in this release.
Fresenius SE & Co. KGaA Registered Office: Bad Homburg, Germany / Commercial Register: Amtsgericht Bad Homburg, HRB 11852
Chairman of the Supervisory Board: Wolfgang Kirsch
General Partner: Fresenius Management SE
Registered Office: Bad Homburg, Germany / Commercial Register: Amtsgericht Bad Homburg, HRB 11673 Management Board: Michael Sen (Chairman), Pierluigi Antonelli, Sara Hennicken, Robert Möller, Dr. Michael Moser
Chairman of the Supervisory Board: Wolfgang Kirsch

March 25, 2026
Bad Homburg, Germany
Consolidated Financial Statements and Management Report (IFRS)

Entdecken Sie am heutigen Weltgesundheitstag, wie die Geschäftsbereiche von Fresenius Forschung und Entwicklung, neue Technologien und Patientenversorgung zusammenbringen, um dazu beizutragen, die Behandlungsergebnisse für Patientinnen und Patienten zu verbessern.
Am 7. April ist der internationale Weltgesundheitstag, der 2026 unter dem Motto „Together for health. Stand with science.“ steht. Er stellt die Bedeutung wissenschaftlicher Erkenntnisse und Zusammenarbeit als Basis für Fortschritte in der Medizin und eine bessere Patientenversorgung in den Mittelpunkt.
Diese Fortschritte entstehen nicht zufällig. Sie sind das Ergebnis intensiver Forschung, dem stetigen Streben nach Innovationen und einer engen internationalen Zusammenarbeit von Wissenschaft, Industrie und klinischer Praxis. Diesem Anspruch folgt auch Fresenius. Viele unserer rund 175.000 Mitarbeitenden arbeiten über alle Segmente hinweg täglich daran, wissenschaftliche Erkenntnisse in konkrete Verbesserungen in der Diagnostik, bei Arzneimitteln und in der Patientenversorgung zu übersetzen. Prof. Dr. med. Ralf Kuhlen, Chief Medical Officer Corporate Development bei Fresenius, bringt es auf den Punkt: „Wir sind bestrebt, wissenschaftliche Erkenntnisse in bessere Versorgung umzusetzen – gemeinsam mit wissenschaftlichen Institutionen haben wir das Ziel, Behandlungen effizienter, sicherer, zuverlässiger und für mehr Patientinnen und Patienten zugänglich zu machen.“
Einer dieser Patienten ist Benjamin Kaiser. Als er nach zwei Wochen anhaltender Bauchschmerzen seine Hausärztin aufsucht, rechnet er nicht mit einer Diagnose, die sein Leben innerhalb weniger Stunden grundlegend verändern würde. Doch in den folgenden Tagen bringt die Diagnostik Gewissheit: Es ist ein aggressives B-Zell-Lymphom. Es folgen belastende Untersuchungen und eine erste Immunchemotherapie. Doch nach sechs Zyklen zeigt sich: Der Tumor wächst weiter. Also müssen für den 37-Jährigen weitere Behandlungsmöglichkeiten in Betracht gezogen werden. Eine Standardbehandlung in dieser Situation ist die CAR-T-Zelltherapie. Zu diesem Zeitpunkt unterliegt die Therapie in Deutschland keinen Einschränkungen mehr. Da sie jedoch hoch spezialisiertes Fachwissen erfordert, wird die Therapie in der Regel nur in ausgewählten Kliniken durchgeführt; ein Grund, warum Benjamin Kaiser an das spezialisierte Behandlungszentrum am Helios Klinikum Berlin-Buch überwiesen wird.
Hier in Berlin-Buch hat Benjamin Kaiser Zugang zu einer CAR-T-Zelltherapie, einer personalisierten Immuntherapie, bei der körpereigene Immunzellen genetisch verändert werden, um Krebszellen gezielt anzugreifen. Bei der Herstellung dieser Therapien können die Zellverarbeitungssysteme LOVO und Cue von Fresenius Kabi eine wichtige Rolle spielen, da sie das Waschen, Konzentrieren, Formulieren und finale Bereitstellen der Zellen automatisieren. Zuvor stabilisiert das Behandlungsteam unter der Leitung von Prof. Dr. Bertram Glaß, Head of Hematology and Oncology am Helios Klinikum Berlin-Buch, den Krankheitsverlauf mit Bestrahlung und Antikörpertherapie und schafft so die Voraussetzung für die Zelltherapie.
Dass diese Therapie heute in Deutschland verfügbar ist, ist das Ergebnis internationaler klinischer Forschung und der aktiven Beteiligung unserer Kolleginnen und Kollegen von Helios. Das Team in Berlin-Buch war an der TRANSFORM-Studie beteiligt und ermöglichte Patientinnen und Patienten früh den Zugang zu dieser Behandlung. Seit 2019 werden hier regelmäßig CAR-T-Zelltherapien durchgeführt. Parallel untersuchen Forschende, wie sich der Einsatz in frühere Krankheitsphasen verlagern lässt, und analysieren europäische Registerdaten, um Therapien gezielter einzusetzen.
Dr. Anna Ossami Saidy, Ärztin in Weiterbildung im Team von Prof. Glaß und Mitglied der europäischen Lymphom-Arbeitsgruppe, analysiert Daten aus dem Register der Europäischen Gesellschaft für Blut- und Knochenmarktransplantation (EBMT). „Erst durch die Auswertung einer großen Anzahl von Patientendaten können wir verstehen, welche Therapie für welchen Patienten am besten wirkt – und Behandlungen gezielt weiterentwickeln“, sagt sie. „Dafür brauchen wir aber immer die Kooperation der Patienten!“ Ihre Ergebnisse zeigen: Verschiedene CAR-T-Zellprodukte sind in vergleichbaren Fällen ähnlich wirksam – ein wichtiger Schritt hin zu individualisierten Therapieentscheidungen. Das Projekt wurde auf dem EBMT-Kongress 2026 ausgezeichnet.
Für Benjamin Kaiser hat diese Forschung unmittelbare Folgen: Heute sind keine Tumorreste mehr nachweisbar. Er plant die Rückkehr in seinen Beruf, weiß aber, dass die Nachsorge entscheidend bleibt. Seine Bereitschaft, Daten für die Forschung zur Verfügung zu stellen, ist für ihn selbstverständlich. Eines wird deutlich: Fortschritt entsteht nicht nur im Labor, sondern auch durch Patientinnen und Patienten, die sich daran beteiligen.
Während auf Intensivstationen täglich Entscheidungen getroffen werden, die das Wohlergehen von Patientinnen und Patienten betreffen, rückt bei Fresenius ein weiterer Faktor in den Fokus: die klinische Ernährung kritisch kranker Patientinnen und Patienten. Hier setzen Initiativen von Fresenius Kabi an – getragen von Forschenden und medizinischen Expertinnen und Experten, die das Ziel verfolgen, die Versorgung durch wissenschaftliche Erkenntnisse weiterzuentwickeln.
Fresenius Kabi investiert in die nächste Generation Forschender. Im Rahmen unserer internationalen JUMPstart-Programme unterstützen wir junge Ärztinnen und Ärzte sowie Wissenschaftlerinnen und Wissenschaftler weltweit mit Mentoring und Schulungen. Ziel ist die Entwicklung eigener Studien – von der ersten Idee bis zur Umsetzung. So entsteht ein wachsendes Netzwerk engagierter Forschender, das neue Impulse für die medizinische Ernährung setzt, etwa in der Intensivmedizin oder bei chronischen Erkrankungen.
Darüber hinaus unterstützen Teams von Fresenius Kabi aus dem Bereich Medical Clinical & Scientific Affairs in Zusammenarbeit mit der European Society of Intensive Care Medicine (ESICM) konkrete Forschungsprojekte in der Ernährungstherapie. Mit dem ESICM-Fresenius Kabi Medical Nutrition Research Grant werden Studien finanziert, die sich mit zentralen Versorgungsfragen befassen, wie beispielsweise Stoffwechsel, Nährstoffbedarf und wirksamen Ernährungsstrategien für kritisch kranke Patientinnen und Patienten. Fellowships und Schulungsprogramme unterstützen junge Intensivmedizinerinnen und -mediziner, neue Erkenntnisse direkt in den klinischen Alltag zu übertragen.
Die beiden Programme greifen ineinander: Während die ESICM-Kooperation aktuelle Forschung in die Anwendung bringt, schafft JUMPstart die Grundlage für die Versorgung von morgen. Beide Initiativen haben das Ziel, wissenschaftliche Erkenntnisse zu erlangen, sie schneller in die klinische Praxis zu überführen und damit die Versorgung kritisch und chronisch kranker Menschen zu verbessern.
Zudem zeigen Initiativen wie das EASYGEN-Konsortium, wie die Zusammenarbeit von Industrie, Kliniken und Wissenschaft neue Therapieansätze vorantreiben kann, beispielsweise indem daran gearbeitet wird, eine dezentrale Herstellung von CAR-T-Zelltherapien zu ermöglichen und den Zugang zu diesen Therapien für Patientinnen und Patienten zu verbessern. „Fresenius Kabi unterstützt die EASYGEN-Initiativen durch die Entwicklung und den Einsatz der nächsten Generation des Cue Zellverarbeitungssystems, das eine neuartige Methode der T-Zellenselektion für die in sich geschlossene Herstellung patienteneigener CAR-T-Zellen in großen Mengen nutzt. Dadurch wird eine duale Systemintegration ermöglicht, die durch die Zusammenarbeit mit TQ Therapeutics vorangetrieben wird“, erklärt Paige Bothwell, Forschungswissenschaftlerin bei Fresenius Kabi Research Technologies. Das Konsortium ist ein Zusammenschluss von 18 Organisationen und umfasst – neben Fresenius Kabi – Fresenius, Helios und Quirónsalud als aktive Partner.
Parallel dazu konzentrieren sich die Aktivitäten von Fresenius Kabi im Bereich Pharma auf die Bereitstellung von lebenswichtigen Medikamenten. Dazu gehören i. v. Arzneimittel und Infusionslösungen, die ein wesentlicher Bestandteil vieler Behandlungsprotokolle sind, insbesondere in der Onkologie und der Intensivmedizin.
Ein zentraler Pfeiler unseres Geschäftsbereichs Biopharma ist die Entwicklung von Biosimilars – biologischen Arzneimitteln, die in Bezug auf Qualität, Sicherheit und Wirksamkeit den Referenzprodukten entsprechen und gleichzeitig dazu beitragen, die Kosten in der Gesundheitsversorgung zu senken. Durch ein vertikal integriertes Modell von der Forschung und Entwicklung bis zum Vertrieb möchte Fresenius Kabi seine Biosimilar-Versorgungskette widerstandsfähig und zuverlässig machen. Auf diese Weise wird wissenschaftliche Innovation in einen breiteren Zugang zu wirksamen Therapien und in nachhaltigere Gesundheitssysteme umgesetzt.