Overview
Financial Results
Q1/2026: REJUVENATE in Action – Demonstrating continued strength and resilient performance
Outlook
FY/26 guidance reconfirmed: Fresenius outlook at a glance.
Financial Reports
Discover Fresenius financial reports and presentations here!
Goals & Strategy
#FutureFresenius is clearly paying off. Find out more about our company’s strategy!

"Fresenius made an excellent start to 2026, delivering performance fully in line with our expectations and leading us to reconfirm our full‑year guidance. In an environment where volatility has become the norm and markets are increasingly focused on earnings visibility and balance sheet discipline, this quarter shows that Fresenius is better prepared than ever. Rejuvenate is working in practice: disciplined execution across our businesses is driving double-digit Core EPS growth, continued margin improvement and a balance sheet that reinforces our financial flexibility. Importantly, we achieved this while continuing targeted investment in innovation and digitalisation – from new product launches to AI-enabled hospital platforms – reinforcing our long-term competitive position. With a more focused organisation and a disciplined financial framework, we are well-positioned to navigate policy and macroeconomic uncertainty while maintaining earnings visibility and delivering sustainable long-term value to patients, partners, and shareholders."
Michael Sen, Chairman of the Management Board
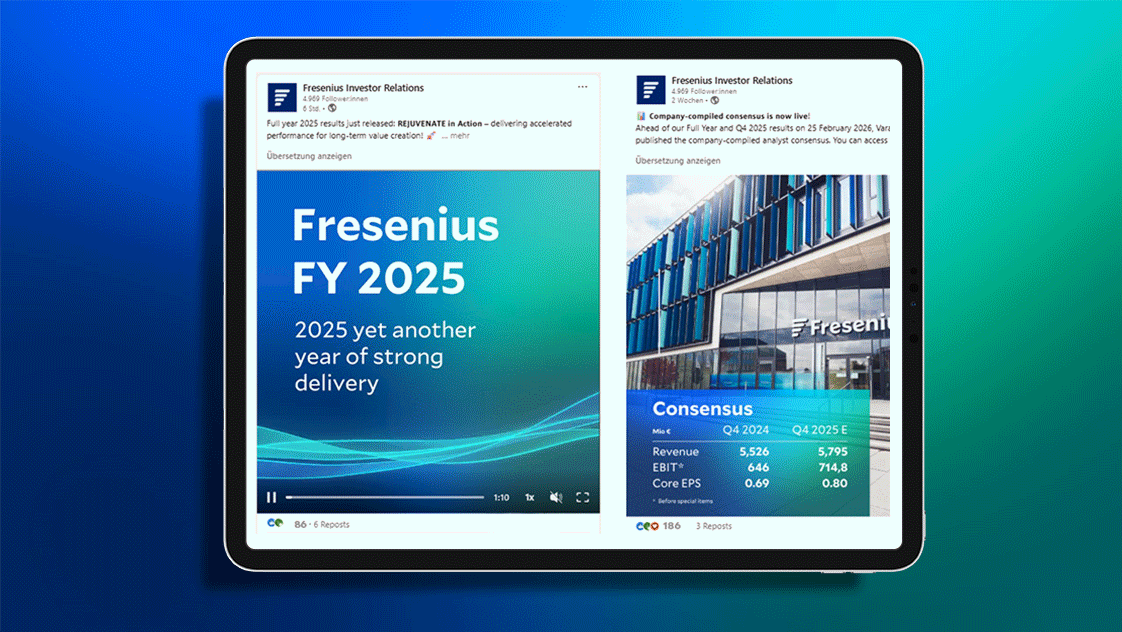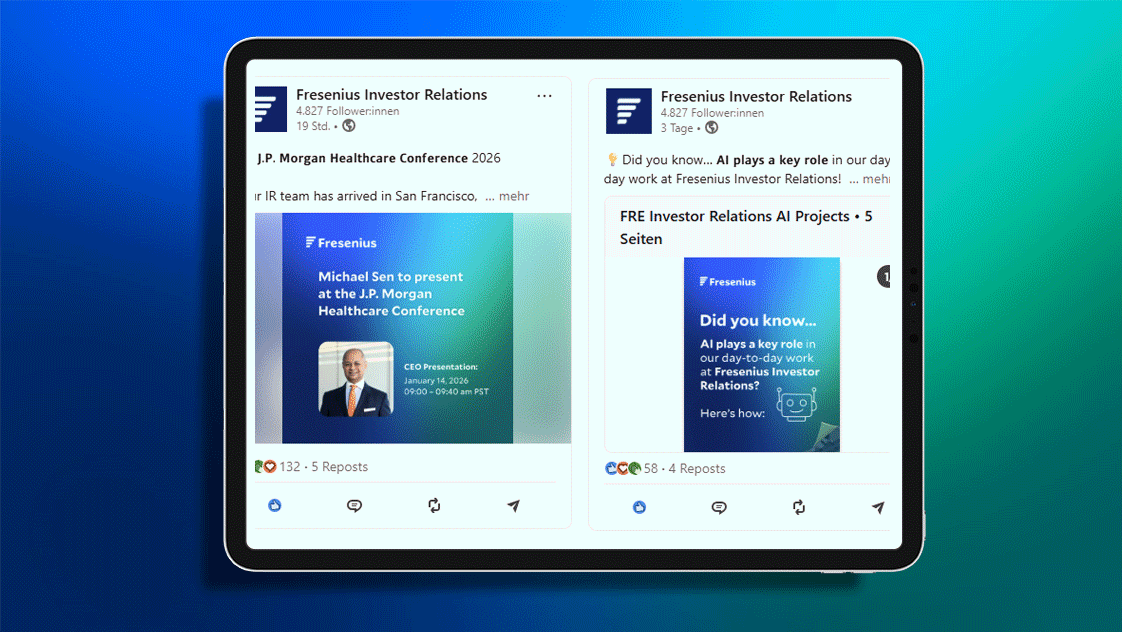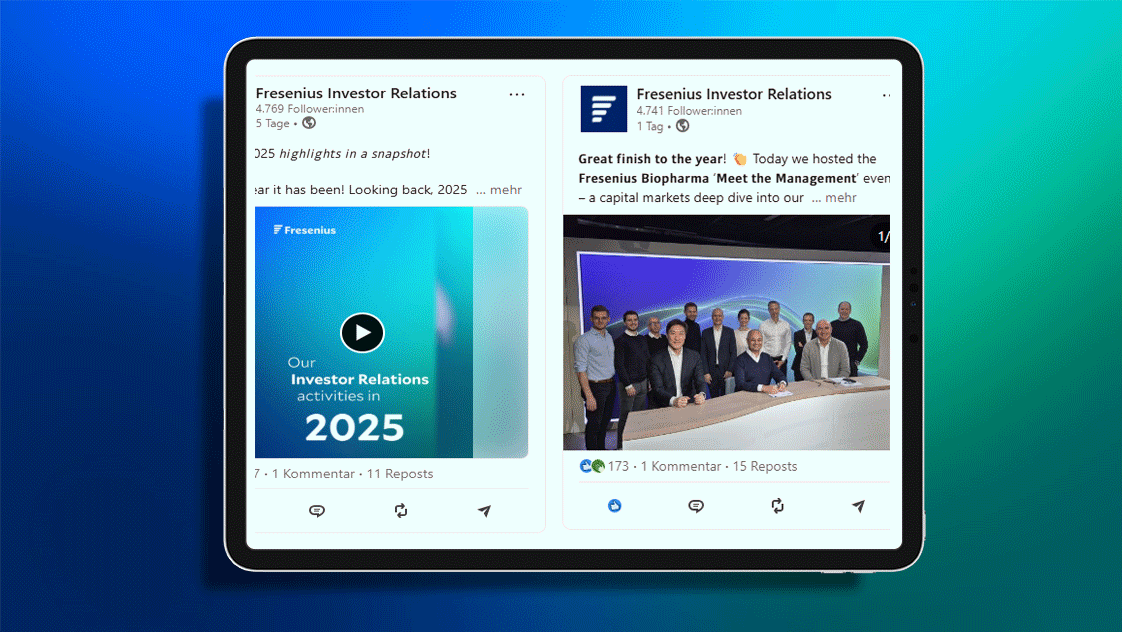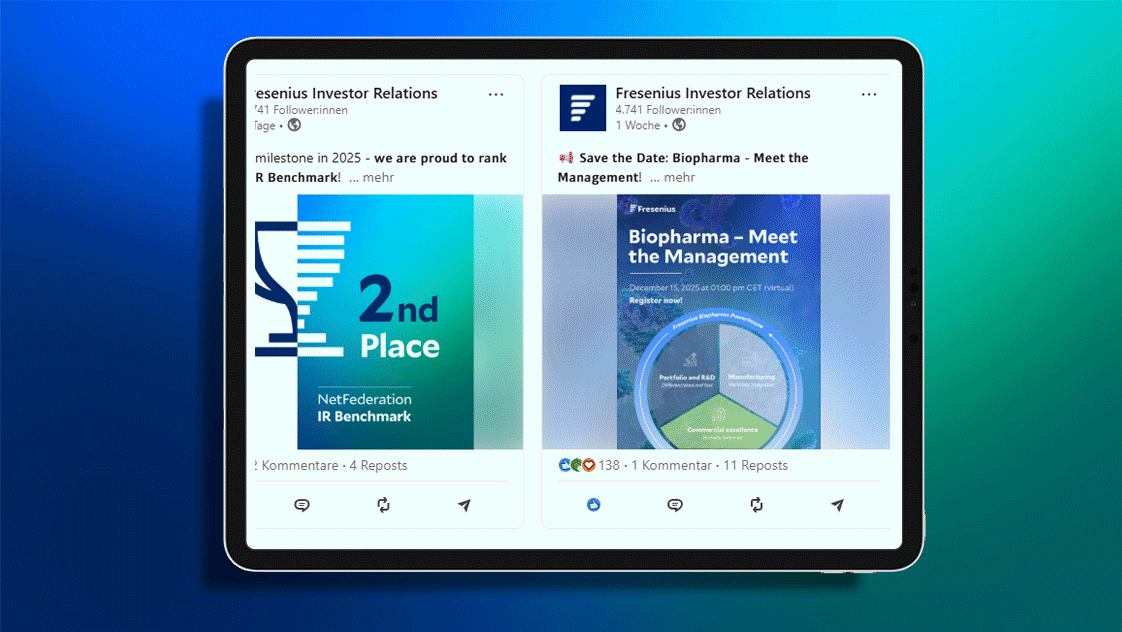
Financial Highlights Q1/2026
Group Revenue 1
€ 5744 m
+5% 2
Q1/25: €5,631 m
Group EBIT 1
€ 678 m
+6% 4
Q1/25: €654 m
Core Net income 1, 3
€ 460 m
+13% 4
Q1/25: €416 m
Core EPS 1, 3
€ 0.82
+13% 4
Q1/25: €0.74
KABI Revenue 1
€ 2150 m
+6% 2
Q1/25: €2,146 m
HELIOS Revenue 1
€ 3501 m
+3% 2
Q1/25: €3,394 m
1 Before special items
2 Organic growth rate adjusted for accounting effects related to Argentina hyperinflation
3 Excluding Fresenius Medical Care and Vitrea
4 Constant currency growth
Letter to our shareholders
Over the past three years, we have successfully implemented our #FutureFresenius strategy and created a new Fresenius that is innovative, relevant, resilient, and adaptable. Read the full CEO-letter!
Sustainability Statement
Our Sustainability Statement shows how we address environmental, social, and governance topics and manage the associated impacts, risks, and opportunities. Read more here!
Strategy and goals
Our vision is to be the trusted, market-leading healthcare company that unites cutting-edge technology and human care to shape next-level therapies. Read more about our corporate strategy!
Rejuvenate
In 2025, Fresenius entered the next phase of our #FutureFresenius strategy: Rejuvenate. We have defined three priorities that guide our actions across the entire company. Discover more in the stories.

Investor Feedback
Open dialogue is valuable for us.
You can submit your feedback quickly and easily using the link below (anonymously if you prefer).
You will be redirected to QuantiFire which conducts this survey in own controllership.
Stay Connected with Fresenius Investor Relations on LinkedIn
Follow Fresenius Investor Relations for expert insights and latest news that contribute to the Fresenius equity story, and exclusive content from our team. Join our growing community and be part of the conversation!