Fresenius Investor Presentation
Fresenius Investor Presentation
With our interactive tool you can analyze and graph a variety of key figures of the Fresenius Group and the business segments on an annual basis.
Contact

Vice President Investor Relations
Deputy Head of Investor Relations
T: +49 (0) 6172 608-5167
florian.feick@fresenius.com
With our interactive tool you can analyze and graph a variety of key figures of the Fresenius Group and the business segments on an annual and quarterly basis.
Contact

Vice President Investor Relations
Deputy Head of Investor Relations
T: +49 (0) 6172 608-5167
florian.feick@fresenius.com
Related Links
Alternative Performance MeasuresTrends towards a changing geopolitical order have been observable and are still continuing since the beginning of the 2025 fiscal year. The potential implications of this for customs duties, taxes, regulation, administration and political decision-making, for example, may have direct and indirect negative effects on the industry environment and the business activities of the Fresenius Group, although these cannot be estimated at present.
Fresenius will continue to closely monitor the potential impact of increased volatility and reduced visibility on its business and balance sheet.
All of these assumptions are subject to considerable uncertainty. Assumptions to guidance: The company acknowledges that the prevailing trends of fast-moving macro-economic and geopolitical environment continue, resulting in increased volatility and a higher level of operational uncertainty. Fresenius’ guidance continues to reflect current factors and known uncertainties such as impacts from tariffs to the extend they can currently be assessed. The guidance does not take into account potential extreme scenarios that could affect the company, its peers, and the healthcare sector as a whole. Potential implications of the United States Supreme Court ruling as of February 20, 2026, are currently being evaluated but cannot be fully assessed at this stage and are hence not reflected in FY/26 guidance.
| Fiscal year 2025 (base) | Targets 2026 | |
|---|---|---|
Fresenius Group | ||
Revenue growth (organic)2 | €22,554 m | 4–7% |
Core EPS growth cc3 | €2.87 | 5–10% |
Operating Companies | ||
Fresenius Kabi | ||
Revenue growth (organic)2 | €8,612 m | Mid-to high-single-digit percentage range |
EBIT margin1 | €1,413 m | 16.5% to 17.0% |
Fresenius Helios | ||
Revenue growth (organic) | €13,550 m | Mid-single-digit percentage range |
EBIT margin1 | €1,328 m | 10.0 to 10.5% |
1 Before special items
2 Organic growth rate adjusted for accounting effects related to Argentina hyperinflation.
3 Excluding Fresenius Medical Care
As of February 25, 2026
Related Links
Interactive Tool#CommittedToLife: the health and well-being of patients is Fresenius’ top priority. For more than 100 years, we have been combining cutting-edge technology with a focus on patients, paving the way for the therapies of the future. We save and improve lives and health. We provide access to affordable and innovative medical products and clinical care of the highest quality.
The global healthcare sector is growing and changing rapidly in response to critical challenges driven by demographic shifts, technological advancements and evolving patient needs. These secular growth drivers are here to stay:
Fresenius is part of the solution: We are a relevant, system-critical healthcare company combining innovative medicine, advanced medical technology, and the highest quality in clinical care to shape next-level therapies.
The healthcare sector is one of the world’s largest industries, offering exceptional growth opportunities. We geared our businesses towards these driving forces and are committed to staying at the forefront, ensuring our businesses remain system-relevant and future-ready.
Our unique strengths lie in our positioning: we cover the entire patient journey with a broad portfolio range – right at the critical spots in care – capable of advancing therapies on many levels. With Fresenius Kabi and Fresenius Helios, we address, lead and meet evolving patient needs across three therapy platforms:
With this strategy, we address the driving forces in healthcare that will change the pattern of growth. All business at Fresenius Kabi and Fresenius Helios have leading market positions in attractive markets and grow meaningfully organically. We are part of the solution of innovative healthcare and future therapies:
Since launching #FutureFresenius in October 2022, we have driven structural productivity and improved returns, creating a simpler, more focused, and stronger healthcare company – delivering clear impact for all stakeholders! Key milestones include deconsolidating Fresenius Medical Care, concluding several important strategic portfolio measures, and exiting Vamed.
In 2025, we kicked-off the next phase of #FutureFresenius - Rejuvenate – taking our performance to the next level! In this phase we focus on:
This means bringing new products and innovations to the market, focusing on the needs of patients and customers. We are investing in AI and digital transformation to enhance clinical decision-making, streamline workflows, and improve patient experiences. These next-generation capabilities will strengthen our leadership in medical quality and innovation.
Overview #FutureFresenius strategy:
Contact

Senior Vice President Investor Relations
Head of Investor Relations
T: +49 (0) 6172 608-97033
nick.stone@fresenius.com
Related Links
Interactive Tool
Discover why investing in Fresenius means investing in long-term healthcare innovation. Fresenius is a relevant, system-critical healthcare company. We combine innovative medicine, advanced medical technology, and the highest quality in clinical care to shape next-level therapies.
Watch the video for a quick summary of the key reasons to invest in Fresenius.
Across our therapy platforms, we deliver real impact for patients, professionals, and hospitals around the world. At the same time, we create long-term value for our shareholders.
Explore below a detailed overview of why Fresenius is both – a system-critical healthcare provider and a well-positioned company.
Global healthcare systems are facing critical challenges:
Fresenius is part of the solution: We are actively addressing these global gaps by:
Our unique strength lies in the strong balance between two system-critical businesses: Fresenius Kabi and Fresenius Helios:
Both businesses hold leading market positions in attractive segments and are set for strong, sustainable organic growth and margin expansion.
In October 2022, Fresenius launched its #FutureFresenius strategy. Since then, we have made significant financial and structural progress, making our company more focused, adaptive, and performance driven.
All key financial metrics in Full-Year 2024 compared to Full-Year 2022 improved – a clear sign that our strategy is working.
Visit our Fresenius Annual Report 2024 for more information.
Persistent macroeconomic volatility and emerging geopolitical tensions, Fresenius delivers consistent performance. This reflects the operating strength and continued execution of #FutureFresenius. While serving global markets, we foster a strong local presence. This way we are able to act quickly – also in uncertainties:
We kicked-off the next phase of #FutureFresenius – Rejuvenate – in 2025: Taking our performance to the next level!
Together they offer strong potential for future growth, innovation and digitalization and thus elevating performance.
Fresenius has transformed into a more focused, performance-driven organization with a strong culture of accountability. Our mission stand firm: to save and improve human lives with affordable, accessible, and innovative healthcare products and the highest quality in clinical care.
With over 100 years of experience, Fresenius has grown from a small business into a global healthcare company. Through #FutureFresenius, we’ve become simpler, more adaptive, and more competitive - delivering real impact to patients, caregivers, hospitals, employees, and shareholders alike. We are ready to lead and remain an employer of choice in today’s competitive healthcare landscape.
Contact

Senior Vice President Investor Relations
Head of Investor Relations
T: +49 (0) 6172 608-97033
nick.stone@fresenius.com
Letter to our shareholders
Over the past three years, we have successfully implemented our #FutureFresenius strategy and created a new Fresenius that is innovative, relevant, resilient, and adaptable. Read the full CEO-letter!
Sustainability Statement
Our Sustainability Statement shows how we address environmental, social, and governance topics and manage the associated impacts, risks, and opportunities. Read more here!
Strategy and goals
Our vision is to be the trusted, market-leading healthcare company that unites cutting-edge technology and human care to shape next-level therapies. Read more about our corporate strategy!
Rejuvenate
In 2025, Fresenius entered the next phase of our #FutureFresenius strategy: Rejuvenate. We have defined three priorities that guide our actions across the entire company. Discover more in the stories.

"2025 was a pivotal year for Fresenius. With disciplined execution of our #FutureFresenius strategy and a strong performance from Team Fresenius, we met our upgraded full-year guidance by delivering another quarter of competitive growth [...]. 2025 capped a year of continued momentum across the Company: We further strengthened the balance sheet, and upgraded our guidance, while preparing the business through targeted investment for the next phase of growth. All of this leads to a proposed dividend of €1.05 per share, underscoring our commitment to creating shareholder value [...]. With #FutureFresenius we have transformed our Company, positioning ourselves to deliver future success in a new world order. Looking ahead, we enter 2026 with strong foundations and clear priorities [...]."
Michael Sen, Chairman of the Management Board
Group Revenue 1
€ 22554 m
+7% 2
FY/24: €21,526 m
Group EBIT 1
€ 2595 m
+6% 3
FY/24: €2,489 m
Net income 1, 4
€ 1619 m
+12% 3
FY/24: €1,461 m
EPS 1, 4
€ 2.87
+12% 3
FY/24: €2.59
KABI Revenue 1
€ 8612 m
+7% 2
FY/24: €8,414 m
HELIOS Revenue 1
€ 13550 m
+7% 2
FY/24: €12,739 m
1 Before special items
2 Organic growth rate adjusted for accounting effects related to Argentina hyperinflation
3 Growth rate at constant currency (cc) and adjusted for Argentina hyperinflation
4 Excluding Fresenius Medical Care
Financial Results
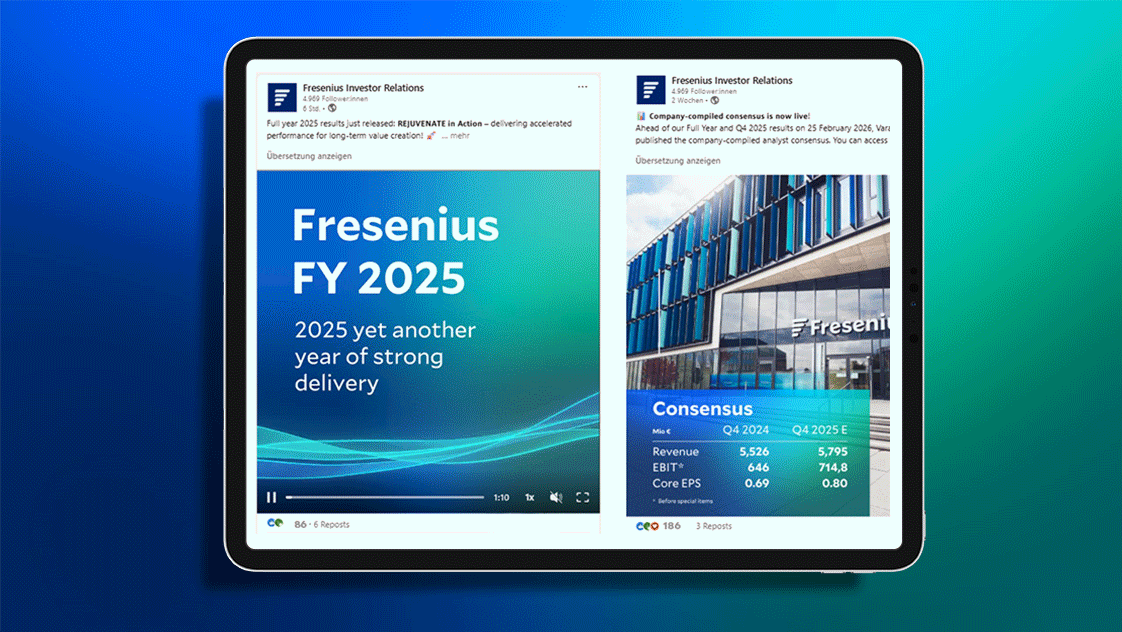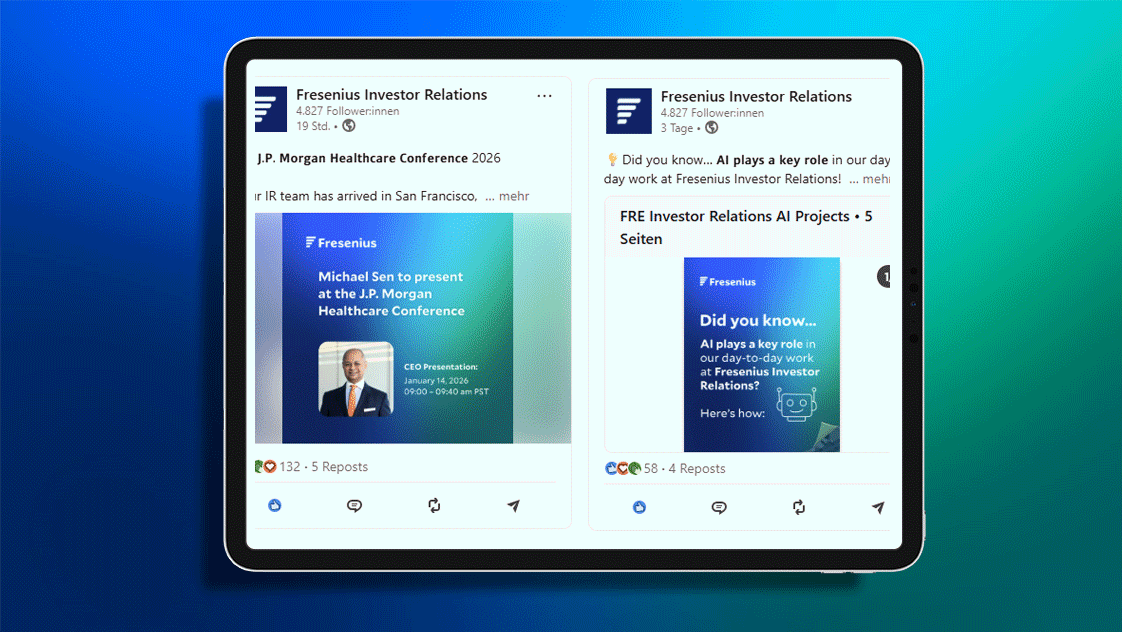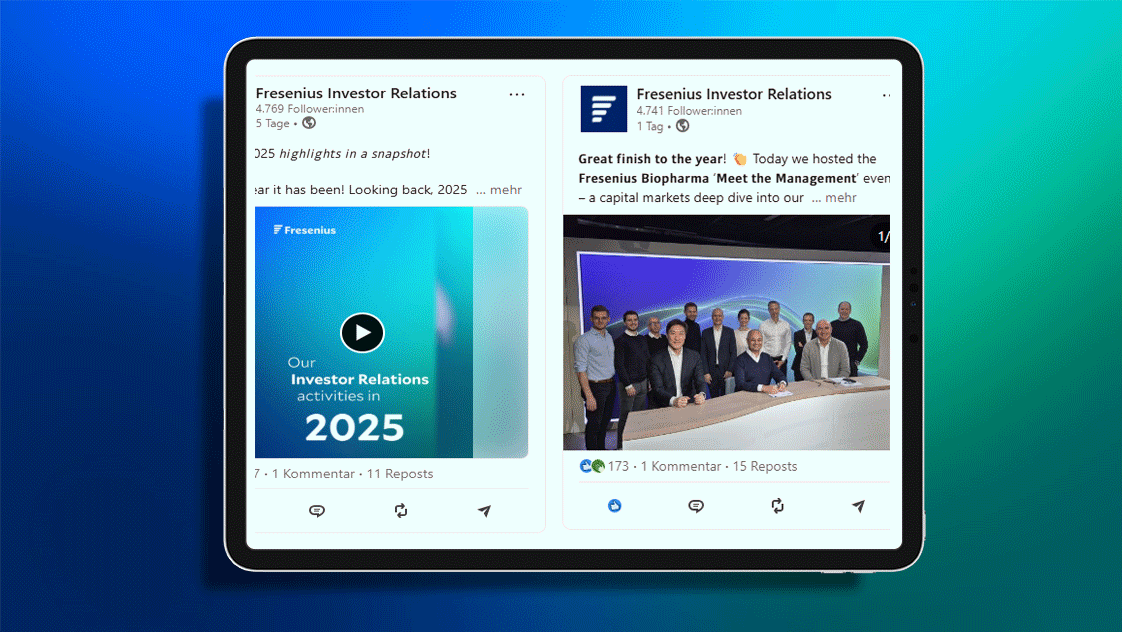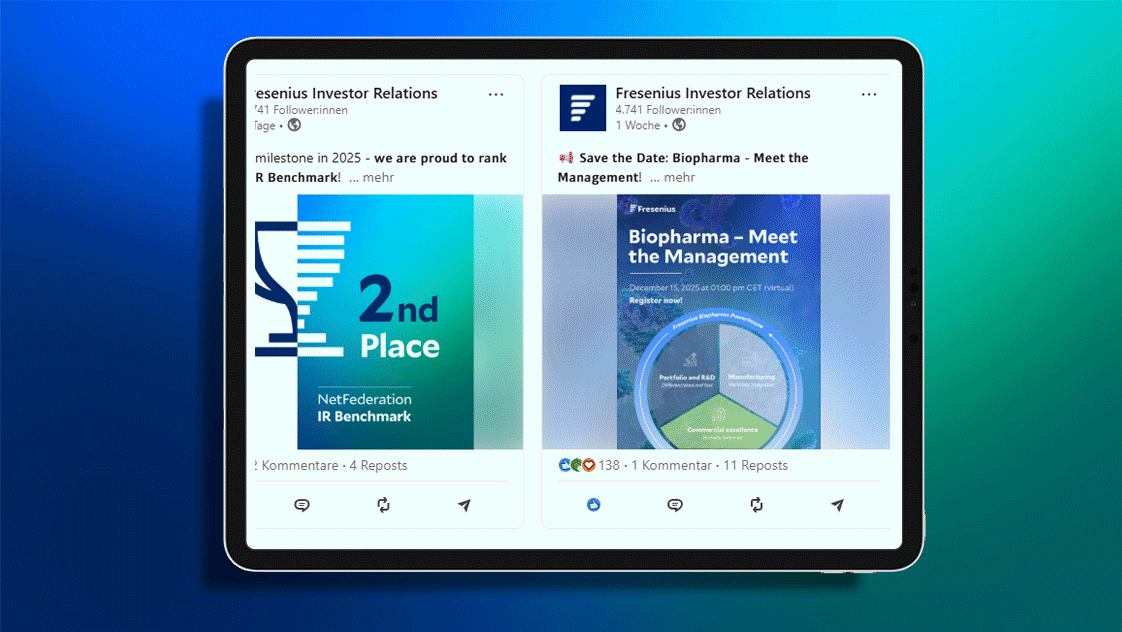
FY 2025: REJUVENATE in Action – Delivering accelerated performance for long-term value creation!
Outlook
New guidance metric established: Fresenius Outlook 2026 at a glance.
Financial Reports
Discover Fresenius financial reports and presentations here!
Goals & Strategy
#FutureFresenius is clearly paying-off. Find out more about our company's strategy!

Open dialogue is valuable for us.
You can submit your feedback quickly and easily using the link below (anonymously if you prefer).
You will be redirected to QuantiFire which conducts this survey in own controllership.
Follow Fresenius Investor Relations for expert insights and latest news that contribute to the Fresenius equity story, and exclusive content from our team. Join our growing community and be part of the conversation!
Fresenius Investor Presentation
Fresenius Investor Presentation
Corporate governance covers all aspects connected with the management, supervision and transparency of companies. Key elements of good corporate governance are efficient company management, the protection of shareholders' interests and transparent corporate communication.
The Supervisory Board of Fresenius SE & Co. KGaA and the Management Board of the general partner of Fresenius SE & Co. KGaA, Fresenius Management SE, issued the Declaration of Conformity pursuant to Section 161 of the German Stock Corporation Act in December 2025.
Current Declaration of Conformity
German Corporate Governance Code
Previous Declarations of Conformity
Please find below the 2024 Corporate Governance Declaration and Report of Fresenius SE & Co. KGaA.
The Management Board takes diversity into account when filling executive positions. At Fresenius, the individual’s qualifications are the paramount consideration in all hiring and promotion decisions. This means that women and men with comparable qualifications and suitability have the same career opportunities. Fresenius will continue to consistently act upon this principle – in compliance with the obligations arising from the Act on the Equal Participation of Women and Men in Leadership Positions in the Private Sector and the Public Sector (FüPoG I) and the Act to Supplement and Amend the Regulations for the Equal Participation of Women in Leadership Positions in the Private Sector and the Public Sector (FüPoG II):
For the Supervisory Board of Fresenius SE & Co. KGaA, the law requires a quota of at least 30% women and 30% men. These mandatory quotas were again met by the Supervisory Board elections in 2021.
The legally stipulated targets for the Management Board do not apply to Fresenius Management SE or to Fresenius SE & Co. KGaA. Due to its legal form, Fresenius SE & Co. KGaA does not have a Management Board. Fresenius Management SE is not listed on the stock exchange and is also not subject to co-determination. In accordance with the legal requirements, the Management Board specifies composition of the two management levels directly below the Management Board as follows:
The first management level includes all Senior Vice Presidents and Vice Presidents who have an employment contract with Fresenius SE & Co. KGaA and who report directly to a Member of the Management Board. Through a decision effective January 1, 2021 the Management Board has set a target, which has to be met by December 31, 2025, and calls for a proportion of women of 30.0% at the first management level.
The second management level includes all Vice Presidents who have an employment contract with Fresenius SE & Co. KGaA and who report directly to a member of the first management level. Through the decision effective January 1, 2021, the Management Board has set a target, which has to be met by December 31, 2025, and calls for a proportion of women of 30.0% at the second management level. The Management Board believes that inclusion in the company-wide long-term incentive programs is a strong indicator that an individual holds a leading executive position. The proportion of women in this group of our top 1,800 executives was approximately 34% as of December 31, 2022.
For a detailed report on the Management and Supervisory Board members’ compensation please refer to the Compensation Report 2024.
For further information regarding the Compensation System 2023+, please refer to the following document:
Our corporate philosophy "entrepreneur in the enterprise" implies not only that our employees are driven by a spirit of entrepreneurial responsibility but that they also have a stake in the company's performance. The overall remuneration package for Group executives and the members of the Management Board has been linked to the performance of the share price through stock option plans.
As of December 31, 2022, Fresenius SE & Co. KGaA had two share-based compensation plans in place: the Fresenius SE & Co. KGaA Long Term Incentive Program 2013 (2013 LTIP) which is based on stock options and phantom stocks and the Long Term Incentive Plan 2018 (LTIP 2018) which is solely based on performance shares. Currently, solely LTIP 2018 can be used to grant performance shares.
In 2023, the new employee participation program Fresenius SHARE was introduced. The program applies equally to all employees of the participating companies - whether collectively agreed or non-pay-scale employees, executives as well as trainees and temporary workers. The program consists of two components: On the one hand, participants can annually purchase a share package with ordinary shares of Fresenius SE & Co. KGaA at a significant discount. Secondly, four targets have been set, upon achievement of which a certain amount will be issued in ordinary shares of Fresenius SE & Co. KGaA. In addition to increasing the Group's net income, the targets include the topics of cybersecurity and quality. The first shares will be issued in 2024, corresponding to the achievement of the targets in the 2023 financial year.
Management Board
Fresenius SE & Co. KGaA does not have an own Management Board. The Management Board of the general partner, Fresenius Management SE, is responsible for conducting the business of the KGaA. It formulates strategy, coordinates this with the Supervisory Board of Fresenius SE & Co. KGaA, and sees to its implementation. It is guided solely by the best interests of Fresenius SE & Co. KGaA.
Supervisory Board of Fresenius SE & Co. KGaA
The Supervisory Board of Fresenius SE & Co. KGaA supervises the management of the Company’s business by the general partner and the latter’s Management Board. The Supervisory Board of Fresenius SE & Co. KGaA has 12 members – 6 shareholder representatives and 6 employee representatives It supervises whether corporate decisions are compliant, suitable, and financially sound. The members of the Management Board of the general partner are appointed by the Supervisory Board of Fresenius Management SE, not by the Supervisory Board of the KGaA.
The Supervisory Board of Fresenius SE & Co. KGaA has the following committees:
- Audit Committee
- Nomination Committee
- Joint Committee
- IT Committee
Supervisory Board of Fresenius Management SE
The Supervisory Board of Fresenius Management SE advises and supervises the Management Board in its management of the Company. The Supervisory Board of Fresenius Management SE appoints the members of the Management Board. He consists of six members who are elected by the annual general meeting of Fresenius Management SE Committees.
Current Publications of transactions that are subject to disclosure requirements are listed, together with the information required by law (before July 3, 2016 pursuant to section 15a of the German Securities Trading Act (WpHG); from July 3, 2016 pursuant to Art. 19 of the Market Abuse Regulation).
2026
2025
2024
2023
2022
2021
Transactions reported during the last 12 months are stored on the Federal Financial Supervisory Authority's central database and can be accessed by clicking on the link Bundesanstalt für Finanzdienstleistungsaufsicht (BAFin).
Member of various supervisory bodies
Chairman of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2021
Current term of office: 2025 – 2029


Date of Birth: | March 19, 1955 |
Place of Birth: | Bensberg |
Nationality: | German |
Professional Experience
2002 - 2018 | DZ Bank AG
|
2000 - 2002 | Deutsche Bank AG, Frankfurt Managing Director and Senior Credit Executive of the Corporates and Real Estate Division and CIB Corporate and Investment Bank |
1998 - 2000 | Deutsche Bank, Singapore General Manager and Chief Country Officer |
1981 - 1998 | Deutsche Bank AG, Düsseldorf and Frankfurt Various positions in corporate and investment banking |
Education/Academic career
1977 - 1981 | University of CologneStudies Business Administration (Diplom-Kaufmann) |
1975 - 1977 | Deutsche Bank AG Banking apprenticeship |
Membership of other statutory supervisory boards | |
B. Metzler seel. Sohn & Co. Aktiengesellschaft (Chair) | |
Membership of comparable German or foreign supervisory bodies | |
None |
Full-time Works Council Member
Helios Vogtland-Klinikum Plauen GmbH
Member of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2018
Current term of office: 2025 – 2029

Date of Birth: | October 1, 1958 |
Place of Birth: | Bad Elster |
Nationality: | German |
Professional Experience
since 09/2018 | Chairman of the Group Works Council Helios Kliniken GmbH |
since 2010 | Member of the European Works Council of Fresenius SE & Co. KGaA |
since 2006 | Helios Vogtland-Klinikum Full-time Works Council Member (Chairman) |
2003 - 2006 | Humaine Vogtland-Klinikum 50% Works Council Member (Chairman); 50% Degreed engineer, Department of biomedical engineering |
1990 - 2003 | Vogtland-Klinikum Plauen Degreed engineer
|
1985 - 1990 | Bezirkskrankenhaus Plauen Degreed engineer |
Education/Academic career
1980 - 1985 | Ilmenau University of Technology Engineering degree in Technical Cybernetics and Biomedical Engineering |
Membership of other statutory supervisory boards | |
Helios Vogtland-Klinikum Plauen GmbH (Fresenius Group mandate) | |
Membership of comparable German or foreign supervisory bodies | |
None |
Member of various supervisory bodies
Deputy Chairman of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2015
Current term of office: 2025 – 2029

Date of Birth: | December 23, 1954 |
Place of Birth: | Bielefeld |
Nationality: | German |
Professional Experience
2003 - 2015 | Allianz SE (formerly Allianz AG) Chairman of the Management Board |
1998 - 2003 | Allianz AG Member of the Management Board |
1988 - 1998 | Allianz Versicherungs-AG |
1983 - 1988 | Diekmann / Thieme GbR (Publishing House) CEO |
Education/Academic career
1973 - 1982 | University of Göttingen Studies of Law and Philosophy |
Membership of other statutory supervisory boards | |
Allianz SE1 (Chair) | |
Membership of comparable German or foreign supervisory bodies | |
None |
1 Stock listed company
Secretary of the Trade Union ver.di Vereinte Dienstleistungsgewerkschaft
Deputy Chair of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2020
Current term of office: 2025 – 2029

Date of Birth: | January 24, 1973 |
Place of BIrth: | Cottbus |
Nationality: | German |
Professional Experience
since 1991 | Trade Union Vereinte Dienstleistungsgewerkschaft ver.di
|
Education
1989 - 1991 | Lausitzer Braunkohle AG Apprenticeship as business management assistant |
Membership of other statutory supervisory boards | |
None | |
Membership of comparable German or foreign supervisory bodies | |
None |
Full-time Works Council Member
Helios Clinic Herzberg and Osterode GmbH
Member of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2025
Current term of office: 2025 – 2029

Date of Birth: | August 03, 1963 |
Place of Birth: | Northeim |
Nationality: | German |
Professional Experience
since 2021 | Member of the European Works Council of Fresenius SE & Co. KGaA
|
Since 2019 | Deputy Chair of the Group Works Council Helios Kliniken GmbH |
since 2014 | Helios Clinic Herzberg and Osterode GmbH
|
1998 – 2014 | Specialist nurse (from 1999) for nursing care in the surgical service Klinik Herzberg and Osterode GmbH (Rhön) |
1991 – 1998 | Nurse in the OR
|
4/83 – 03/91 | Temporary soldier
|
Education
1983 – 1986 | Training as a nurse |
1997 – 1999 | Training as a specialist nurse for nursing in the surgical service |
Membership of other statutory supervisory boards | |
None | |
Membership of comparable German or foreign supervisory bodies | |
None |
Chairman of the Board and Medical Director
of the Mainz University Medical Center
Member of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2025
Current term of office: 2025 – 2029

Date of Birth: | May 14, 1970 |
Place of Birth: | Wiesbaden |
Nationality: | German |
Professional Experience
since 2024 | Mainz University Medical Center Chairman of the Board and Medical Director |
2021 – 2023 | Helios Dr. Horst Schmidt Kliniken Wiesbaden Medical Director |
2014 – 2021 | Helios Dr. Horst Schmidt Kliniken Wiesbaden Gastroenterology Clinic Director, Medical Director |
2012 – 2014 | St. Marienkrankenhaus Frankfurt Chief Physician for Internal Medicine and Gastroenterology |
2008 – 2012 | Mainz University Medical Center W2 Professorship for Gastrointestinal Endoscopy |
Education/Academic career
2005 | Mainz University Medical Center Habilitation |
1996 | Mainz University Medical Center Promotion |
1990 – 1996 | Mainz University Medical Center Studies of human medicine |
Membership of other statutory supervisory boards | |
None | |
Membership of comparable German and foreign supervisory bodies | |
None |
Full-time Works Council Member
Quirónsalud Hospital Universitari General de Catalunya
Senior trade union secretary of the UGT trade union
for the private healthcare sector Barcelona
Member of the Supervisory Board of Fresenius SE & Co. KGaA
First appointment: 2025
Current term of office: 2025 – 2029
Date of Birth: | May 25, 1984 |
Place of Birth: | Barcelona |
Nationality: | Spanish |
Professional Experience
since 2025 | Member of the European Works Council of Fresenius SE & Co. KGaA |
since 02/2024 | Mediator and Conciliator of the UGT trade union Labor Court of Catalonia Barcelona, Barcelona |
since 06/2020 | General Secretary of the UGT trade union UGT representative for occupational health and safety Quirónsalud Hospital Universitari General de Catalunya San Cugat del Vallés, Barcelon |
since 01/2006 | Administration Emergency Department Quirónsalud Hospital Universitari General de Catalunya San Cugat del Vallés, Barcelona |
Education/Academic career
2008 - 2010 | Master‘s degree in secretarial and operational management Vapor Universitario de Terrassa, Barcelona |
2006 - 2008 | CFGS Administration and Finance IES Terrassa, Barcelona |
2004 - 2005 | CFGM Administrativa IES Santa Eulalia, Barcelona |
1996 - 2000 | Secondary school diploma I.E.S. Cavall Bernat, Barcelona |
Membership of other statutory supervisory boards | |
None | |
Membership of comparable German and foreign supervisory bodies | |
None |
Member of various supervisory bodies
Member of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2016
Current term of office: 2025 – 2029

Date of Birth: | November 17, 1960 |
Place of Birth: | Offenbach / Main |
Nationality: | German |
Professional Experience
2008 - 2024 | UCB S.A. Chief Medical Officer and Executive Vice President Development |
2001 - 2009 | Schwarz Pharma AG Member of the Executive Board, Head of Research and Development |
2000 - 2001 | BASF Pharma Vice President Global Projects |
1992 - 2000 | Hoechst AG Various positions, last serving as Vice President Clinical Development |
Education/Academic career
since 2000 | University of Frankfurt am Main Professor for Internal Medicine |
1985 | University of Frankfurt am Main Doctorate in medicine (Dr. med.) |
1979 - 1985 | University of Frankfurt am Main Studies of medicine |
Membership of other statutory supervisory boards | |
Evotec SE1 (Chair) | |
Membership of comparable German or foreign supervisory bodies | |
Sobi Swedish Orphan Biovitrum1 (Member of the Board of Directors) |
1 Stock listed company
Full-time Works Council Member
Fresenius Kabi Deutschland GmbH
Member of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2023
Current term of office: 2025 – 2029

Date of Birth: | May 18, 1969 |
Place of Birth: | Marburg |
Nationality: | German |
Professional Experience
since 2002 | Fresenius Kabi Deutschland GmbH, Standort Friedberg
|
since 2021 | Honorary Judge, Hessian Tax Court |
since 2017 | IHK-Examiner, IHK Frankfurt |
since 2015 | Honorary Judge, Labour Court |
2007 – 2018 | Laboratoy Manager, IPK-Laboratories |
2003 – 2007 | Quality Control Technician |
1992 – 2003 | Quality Control Laboratory Assistant |
Education
2021 | Specialist for Occupational Health Management, IHK |
2020 – 2021 | Certified Specialist for Data Protection and Data Security |
2018 | Project Manager, IHK |
1999 – 2001 | Training as Bachelor Professional of Management for Industry |
1985 – 1989 | Apprenticeship as Chemical Laboratory Technician |
Membership of other statutory supervisory boards | |
None | |
Membership of comparable German or foreign supervisory bodies | |
None |
Works Council Member
Fresenius Kabi España S.A.U.
Laboratory Technician for the Pharmaceutical Industry
Member of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2016
Current term of office: 2025 – 2029

Date of Birth: | January 29, 1974 |
Place of Birth: | Barcelona |
Nationality: | Spanish |
Professional Experience
since 2020 | Spokesman of the works council for the company equal opportunities and equal opportunities plan (Plan de Igualdad) |
since 2016 | Member of the Supervisory Board of Fresenius SE & Co. KGaA |
since 2008 | Member of the European Works Council of Fresenius SE & Co. KGaA |
since 2004 | Member of the Works Council Fresenius Kabi España S.A.U. |
since 1998 | Laboratory Technician for Pharmaceutical Products at Fresenius Kabi España S.A.U |
Education
Since 2024 | Further education and training on CSRD and sustainability, including analysis of reports on a company‘s environmental, social and ethical performance; Trade union action for ecological transition and energy transition, JMZ Trade Union University |
Since 2020 | Further education and training on the topic of the Gender Equality and Equal Opportunities Plan according to Ley Orgánica 3/2007, with a focus on rules for pay transparency, gender equality audits and reporting obligations |
1998 | Training as a laboratory technician in the pharmaceutical industry Fresenius Kabi España S.A.U. |
1990 - 1994 | Secondary school graduation Instituto Cristófol Ferrer, Secondary school |
Membership of other statutory supervisory boards | |
None | |
Membership of comparable German and foreign supervisory bodies | |
None |
Member of various supervisory bodies
Member of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2022
Current term of office: 2025 – 2029

Date of Birth: | February 28, 1961 |
Place of Birth: | Marl |
Nationality: | German |
Professional Experience
2014 – 2021 | LPEQ Ltd. (Listed Private Equity Association), London Member of the Board, Non Executive Director |
2012 - 2022 | Deutsche Beteiligungs AG, Frankfurt / Main Member of the Executive Board / Chief Financial Officer Responsible for Finance and Accounting, Investor Relations, Legal and Tax, Portfolio Valuation, Risk Management, Internal Audit, Human Resources, Organization, IT (incl. cybersecurity) |
2011 – 2012 | Kirche in Not, Königstein im Taunus Managing Director at the global headquarter of the organization |
1990 – 2011 | KPMG AG, Frankfurt / Main Last position: Partner
|
1987 - 1990 | Winterhager Dr. Heintges Stützel Laubach GmbH, Wirtschaftsprüfungsgesellschaft, Düsseldorf Last position: Auditor and tax specalist |
Education/Academic career
1995 | Appointment as auditor (2013 waiver of appointment) |
1992 | Appointment as tax advisor (2022 waiver of appointment) |
1980 - 1987 | Westfälische Wilhelms-Universität Münster
|
Membership of other statutory supervisory boards | |
Fresenius Management SE (Fresenius Group mandate) | |
Membership of comparable German and foreign supervisory bodies | |
None |
Member of various supervisory bodies
Member of the Supervisory Board of Fresenius SE & Co. KGaA
Initial appointment: 2022
Current term of office: 2025– 2029

Date of Birth: | April 13, 1961 |
Place of Birth: | Stuttgart |
Nationality: | German |
Professional Experience
2018 – 2022 | Siemens Healthineers AG, Erlangen Member of the Managing Board Responsible for Imaging, Advanced Therapies, Technology & Innovation, Quality & Regulatory Affairs, Sustainability |
2018 – 2019 | Siemens Healthineers AG, Erlangen President Business Segment Diagnostic Imaging |
2015 – 2018 | Siemens AG, Healthcare Sector, Erlangen SVP, General Manager of Business Line Magnetic Resonance |
2014 – 2015 | Beckmann-Coulter – A Danaher Company, Miami (USA) SVP, Head of Business Unit Hematology and Urinalysis |
1998 – 2014 | Siemens AG, Healthcare Sector, Knoxville (USA)
|
2012 – 2014 | Siemens AG, Healthcare Sector, Knoxville (USA) CEO PETNET Solutions (Radiopharmaceuticals) |
1994 – 1998 | University of Tübingen Resident Physician Surgery |
1993 – 1994 | University of Frankfurt am Main Resident Physician Internal Medicine, Nuclear Medicine |
Education/Academic career
1995 | University Frankfurt am Main Doctor of Medicine, M.D. (Dr.) |
1985 – 1992 | University Frankfurt am Main Studies in Medicine |
Membership of other statutory supervisory boards | |
Gerresheimer AG1 | |
Membership of comparable German and foreign supervisory bodies | |
None |
1 Stock listed company
Wolfgang Kirsch (Chair)
Michael Diekmann
Susanne Zeidler
Susanne Zeidler (Chair)
Bernd Behlert
Grit Genster
Wolfgang Kirsch
Dr. Christoph Zindel
Dr. Dieter Schenk (Chair)
Michael Diekmann
Wolfgang Kirsch
Susanne Zeidler
The committee consists equally of two members each of the Supervisory Board of Fresenius SE & Co. KGaA and of Fresenius Management SE.
Dr. Christoph Zindel (Chair)
Óscar Romero de Paco
Susanne Zeidler
Rules of Procedure
Rules of Procedure
CVs of the Supervisory Board members
Meeting participation by individual
Chairman of the Supervisory Board of Fresenius SE & Co. KGaA
Former Chief Executive Officer of Deutsche Post DHL Group
Member of various Supervisory Boards
Member of various Supervisory Boards
Deputy Chairman
Member of supervisory bodies
Member of supervisory bodies
Contact