Fresenius Medical Care, the world’s largest provider of dialysis products and services, today announced the closing of the divestiture of Sound Inpatient Physicians Holdings, LLC (Sound) to an investment consortium led by Summit Partners as of June 28, 2018. The total transaction proceeds are $2.15 billion (EUR 1.85 billion1).
The divestment of Sound is aligned with Fresenius Medical Care’s goal of further sharpening the profile of the company’s Care Coordination portfolio. The transaction is expected to generate a positive effect on net income2 for Fresenius Medical Care of around USD 7523 million (EUR 648 million1). This effect includes the half year impact from the increase in valuation of Sound´s share based payment program caused by the divestment of Sound.
The financial targets for 2018 and 2020 do not include the effects of this divestiture. The divestment reduces on a constant currency basis Fresenius Medical Care´s 2018 revenue expectations by around EUR 650 million and net income2 by around EUR 40 million. The growth rates indicated in the financial targets for 2018 will be achieved on a comparable basis4 following the divestiture of Sound.
1 EUR/USD 1.16
2 Net income attributable to shareholders of Fresenius Medical Care AG & Co. KGaA
3 Based on the company´s latest available information
4 On a pro-forma basis revenue for Sound in H2 2017 was around EUR 560 million and net income was around EUR 40 million
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Fresenius Medical Care AG & Co. KGaA does not undertake any responsibility to update the forward-looking statements in this release.
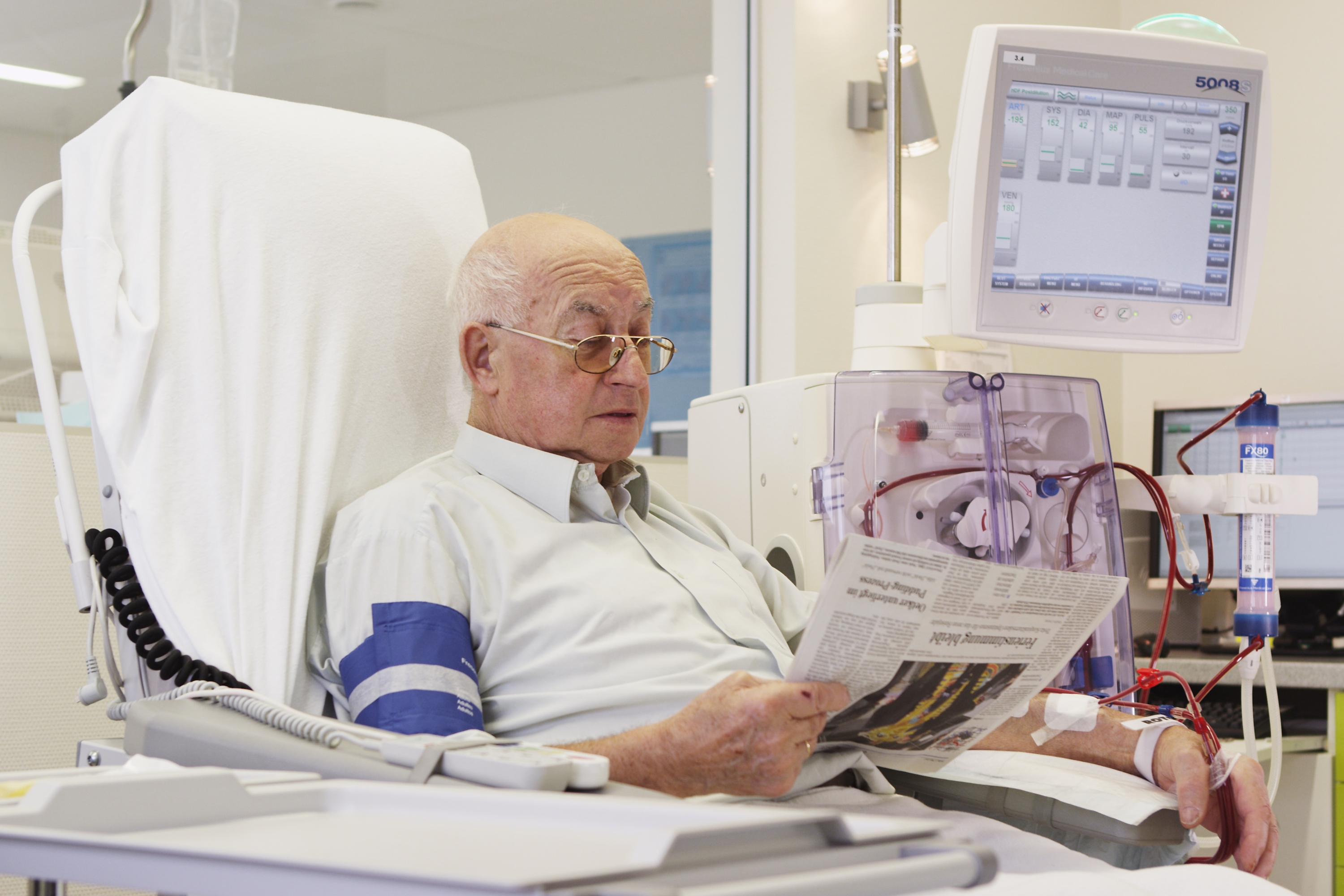
Fresenius Medical Care, the world’s largest provider of dialysis products and services, and Humacyte, Inc., a medical research, discovery and development company, today announced a strategic, global partnership and a $150M USD equity investment. This agreement has the potential to make Humacyte’s investigational human acellular vessel, HUMACYL®, available to more patients worldwide following approval of the product. HUMACYL is currently being investigated for vascular access for hemodialysis and may prove more effective than current synthetic grafts and fistula. Under the terms of the agreement, Fresenius Medical Care will obtain the exclusive global rights to commercialize HUMACYL.
Fresenius Medical Care will be responsible for the marketing, sales and distribution of HUMACYL following approval by the relevant health authorities. In addition, Fresenius Medical Care will make a $150M USD equity investment in Humacyte to gain a 19% fully diluted ownership stake in the company. With the investment, Fresenius Medical Care will have the opportunity to bring transformative clinical innovation in the form of Humacyte’s bioengineered human acellular vessels to the worldwide end stage renal disease (ESRD) patient population following product approval. The transaction is subject to customary closing conditions, including expiration or termination of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended, and is expected to close in July 2018.
“By partnering with Humacyte, Fresenius Medical Care has an opportunity to offer a dialysis vascular access option with the potential for significant clinical efficacy and safety improvements, including the potential to minimize catheter contact time to the benefit of our patients,” said Franklin Maddux, MD, Chief Medical Officer for Fresenius Medical Care North America. “Our exclusive rights to distribute this innovative technology to dialysis patients worldwide may have significant benefits not only to patients, but health systems as well. With the potential for fewer anticipated complications and interventions compared to synthetic grafts, we may see increased safety for patients and reduced medical and economic burdens to the healthcare system.”
The current vascular access modalities necessary to deliver dialysis treatment include fistulas, grafts and central venous catheters. All three options have limitations. Half of fistulas fail and do not mature in patients, delaying vascular access for dialysis treatment. In the meantime, many patients need a central venous catheter, which significantly increases the risk of infection. Humacyte has developed a novel human tissue-based investigational product, HUMACYL, for patients with ESRD requiring hemodialysis. Compared to an arteriovenous fistula, HUMACYL can be available for use in hemodialysis within weeks and may have an overall higher rate of maturation. It also may offer a more durable, biologic alternative to synthetic grafts.
“This is a transformational milestone for Humacyte, giving us the world’s strongest partner to help bring our product to more patients globally,” said Carrie Cox, CEO and Chairman of Humacyte, Inc. “Our partnership will allow Humacyte to focus on advancing the potential for HUMACYL as a substantial breakthrough in the science of regenerative medicine, and to continue our development of an exciting future pipeline.”
Humacyte’s bioengineered blood vessel is currently in Phase III pivotal trials in the U.S. and Europe, and the company plans to seek regulatory approval in both regions upon completion of the trials.
Disclaimer
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, risks and uncertainties in research and development and the regulatory approval process; failure to satisfy the conditions to the consummation of the transaction, including the receipt of regulatory approval; regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Without prejudice to its obligations under capital market laws, neither Fresenius Medical Care AG & Co. KGaA nor Humacyte, Inc. undertakes any responsibility to update the forward-looking statements in this release.
More than a year after closing its acquisition of the Spanish hospital operator Quirónsalud, Fresenius Helios foresees strong prospects for further international growth. At a Capital Markets Day in Berlin today, Fresenius provided investors and analysts with an overview of the progress made in the cooperation between Helios Germany and Quirónsalud, which is opening up new growth opportunities in many areas.
Fresenius Helios confirmed the medium-term target for synergies of €50 million annually. Cooperating in laboratory services and joint purchasing has already achieved cost savings in the current business year that should increase to €30 million per year in the medium term. Annual sales synergies of about €20 million are expected from knowledge transfers in medicine, new models for patient care, and digitalization, among other measures.
Quirónsalud has already begun to implement the system developed by Helios Germany for measuring and assessing data on medical quality. To increase transparency and spur individual hospitals to compete on quality, Helios Germany publishes treatment data on the most important and common medical indications for each hospital in direct comparison with national averages compiled by Germany’s Federal Statistical Office. Quirónsalud has already adapted most of these quality indicators. It has also launched peer reviews, collegial exchanges in which the responsible physicians from the individual hospitals advise and consult with each other on questions concerning treatment quality. These exchanges have already led to significant quality improvements at Helios Germany.
The knowledge transfer also extends to digitalization: Quirónsalud’s advanced know-how in patient-oriented uses includes the development of apps, while Helios Germany is particularly strong in IT processes.
Stephan Sturm, CEO of Fresenius, said: “Helios Germany and Quirónsalud are bundling their respective strengths across national borders, exchanging experience and knowledge. This benefits our patients, in Spain as well as in Germany. And it is creating, step by step, the economic prerequisites for the further internationalization of our hospital business. The driving force behind our business success is, and will remain, our clear focus on the well-being of patients. So wherever and whenever we can do more for our patients through closer cooperation between our business segments, we will seize that opportunity.”
Fresenius Helios will also profit from combining the experience gained from the very different health care systems of Spain and Germany. The Spanish system, for its part, allows a great deal of flexibility in care provision models. In Madrid, for example, Quirónsalud has been given responsibility for providing health care to publicly funded patients in designated parts of the city in return for a set reimbursement rate. For patients, however, treatment quality remains the decisive criterion, and assigned patients are still free to choose another hospital. If they do, the cost must be assumed by the hospital that was originally assigned to their care. This increased competition stimulates continuous improvement in areas that are of crucial importance to patients – such as medical quality, service and shorter waiting times. All of these are core competencies of Quirónsalud.
Another area where Fresenius Helios can now draw on its experience from two different health care systems is in classifications of inpatient and outpatient care. The two are strictly separated in Germany, but less so in Spain: Many treatments and after-care procedures that Germans undergo as inpatients are provided to their Spanish counterparts on an outpatient basis – often resulting in significantly shorter hospital stays.
Dr. Francesco de Meo, who is responsible for Fresenius Helios on the Fresenius Management Board, said: “Helios Germany and Quirónsalud are leaders in their home markets, each of which has different reimbursement and care models and health insurance systems. In response, each company has developed its own strengths, which excellently complement each other and can be used to their mutual advantage. We expect this will contribute to higher medical quality and more efficiency, and bring us closer to patients. Thus, together we are building a common base of knowledge and experience that will help us to enter new markets.”
Cooperation with Fresenius Vamed is also being intensified – for example, in procurement, where Fresenius Helios and Fresenius Vamed are now jointly purchasing certain products. In addition, Fresenius Vamed has started providing Quirónsalud hospitals in Spain with technical services and medical technology, as it was already doing for Helios Germany. In Germany, meanwhile, Fresenius Helios and Fresenius Vamed will combine their know-how in hospital construction in order to bundle their competencies in construction and project management.
Earlier this week, the two companies agreed that Fresenius Helios’ inpatient rehabilitation business in Germany will be transferred to Fresenius Vamed on July 1, 2018. This will put Fresenius Helios on a stronger growth footing, with an even clearer focus on the acute-care hospital business and its further internationalization.
Webcast of the event:
Fresenius Helios Capital Markets Day will be available as a webcast on the Internet under: www.fresenius.com/investors-new-developments
This release contains forward-looking statements that are subject to various risks and uncertainties. Future results could differ materially from those described in these forward-looking statements due to certain factors, e.g. changes in business, economic and competitive conditions, regulatory reforms, results of clinical trials, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. Fresenius does not undertake any responsibility to update the forward-looking statements in this release.

Fresenius is reorganizing the Group’s inpatient rehabilitation business, to create the conditions for the continued growth of Fresenius Helios and Fresenius Vamed. On July 1, 2018, 38 health care facilities and 13 service companies in Germany specializing in inpatient post-acute and nursing care, which are now operated by Fresenius Helios, will be transferred to Fresenius Vamed. This will strengthen Fresenius Vamed’s position as a leading provider of post-acute care in Europe. Fresenius Helios, meanwhile, will focus even more strongly on the acute care hospital business and its continued internationalization.
The transaction has a total volume of €485 million, including assumed net debt of €15 million. It will be financed Group-internally. This year, the inpatient post-acute care business that is being transferred is expected to generate about €460 million in sales and an EBIT of around €37 million.
As part of the transfer, Fresenius Vamed is taking on all of the approximately 7,700 employees of the post-acute care business.
With regard to the respective core competencies of Fresenius Vamed and Fresenius Helios, the two Fresenius business segments will further intensify their cooperation, which has already proven successful in Germany and Spain.
Stephan Sturm, CEO of Fresenius, said: “For Fresenius, post-acute care is and will remain an important component of the treatment we provide to our patients. And now by bundling our great expertise in this area within Fresenius Vamed, we are building a platform for further international growth. We are also putting Fresenius Helios’ on a stronger growth footing, with an even clearer focus on acute care. In addition, we are facilitating even more intensive cooperation between these two business segments, for the benefit of our patients.”
Fresenius Vamed is already a leading post-acute care provider in Austria, Switzerland and the Czech Republic, and entered the British market last year. After the transaction, Fresenius Vamed will have a total of 63 inpatient health care facilities in five European countries.
Even after the transfer of its inpatient post-acute care business to Fresenius Vamed, Fresenius Helios will remain Europe’s largest private hospital operator, with 137 hospitals and about 100,000 employees in Germany and Spain. The strategic focus of Fresenius Helios will remain its acute care hospitals, as well as the outpatient acute care - including preventative medicine - and outpatient post-acute care.
As a consequence of the transfer, Fresenius is adjusting the 2018 outlook for Fresenius Helios and Fresenius Vamed. Fresenius Helios now expects EBIT growth of 5% to 8% (7% to 10% previously). Its organic sales growth outlook of 3% to 6% remains unchanged. Fresenius Vamed is now expecting EBIT growth of 32% to 37%1 (5% to 10% previously). Fresenius Vamed’s organic sales growth forecast remains unchanged at 5% to 10%.
The transaction will not significantly affect the 2018 business results of the Fresenius Group, which accordingly confirms its guidance2 for the current business year. Group sales are expected to increase by 5% to 8%3 in constant currency. Net income4,5 is expected to grow by 6% to 9% in constant currency. Excluding expenditures for the further development of the biosimilars business, net income4,6 is expected to grow by ~10% to 13% in constant currency.
1 Expected EBIT of the inpatient post-acute care business in H2/2018: ~€20 million
2 Excluding effects of the Akorn, NxStage and Sound Physicians transactions
3 2017 base adjusted for IFRS 15 adoption (deduction of €486 million at Fresenius Medical Care)
4 Net income attributable to shareholders of Fresenius SE & Co. KGaA
5 2017 base: €1,816 million; 2018 before special items (i.e., transaction-related effects); including expenditures for further development of biosimilars business (€43 million after tax in FY/17 and ~€120 million after tax in FY/18)
6 2017 base: €1,859 million; 2018 before special items (i.e., transaction-related effects)
This release contains forward-looking statements that are subject to various risks and uncertainties. Future results could differ materially from those described in these forward-looking statements due to certain factors, e.g. changes in business, economic and competitive conditions, regulatory reforms, results of clinical trials, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. Fresenius does not undertake any responsibility to update the forward-looking statements in this release.
Fresenius remains on track to continue strong growth. At the Annual General meeting in Frankfurt today, Stephan Sturm, CEO of Fresenius, confirmed the global health care group’s 2018 targets, which foresee increases of 5 to 8 percent in sales and 6 to 9 percent in net income1,2, both in constant currency. Excluding the investments for the development of the biosimilars business, net income1 is expected to increase by about 10 to 13 percent in constant currency.
“Fresenius has been highly successful over many years. And Fresenius will continue to be highly successful in the coming years,” Sturm said in his speech to shareholders. “Our entry into biosimilars. The expansion of our home dialysis activities. The internationalization of Fresenius Helios. And our huge investments to secure even more quality and efficiency in our production. These are all examples of how we are shaping our business, today, to stay successful in the future. The dynamic growth of Fresenius continues.”
Shareholders approved with a majority of 89.2 percent the proposal of the General Partner and the Supervisory Board to increase the dividend for the 25th consecutive time. It was raised by 21 percent, to €0.75 per share.
With a majority of 94.3 percent, the shareholders approved a new Authorized Capital I in the amount of €125 million. New authorizations to issue convertible bonds, to repurchase own shares, and to use equity derivatives for repurchasing own shares were also approved. In addition, the shareholders approved the revised compensation system for the members of the Management Board of the General Partner.
Shareholder majorities of 97.5 percent and 89.0 percent, respectively, approved the actions of the Management and Supervisory Boards in 2017.
At the Annual General Meeting, 73 percent of the subscribed capital was represented.
1 Net income attributable to shareholders of Fresenius SE & Co. KGaA; 2017 base: €1,859 million; 2018 before special items (i.e., transaction-related effects)
2 including expenditures for further development of biosimilars business (€43 million after tax in FY/17 and ~€120 million after tax in FY/18)
This release contains forward-looking statements that are subject to various risks and uncertainties. Future results could differ materially from those described in these forward-looking statements due to certain factors, e.g. changes in business, economic and competitive conditions, regulatory reforms, results of clinical trials, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. Fresenius does not undertake any responsibility to update the forward-looking statements in this release.
Fresenius Medical Care, the world’s largest provider of dialysis products and services, expects continued strong growth. At the Annual General Meeting in Frankfurt today, CEO Rice Powell explained the company's growth strategy for the coming years: “We will continue to expand our business with services and products for dialysis, and will continue to grow,” Powell said in his speech to the shareholders. “The basis for this is our international network of dialysis centers, our comprehensive knowledge in dialysis, and focusing our Care Coordination portfolio. We are well positioned to respond to the current and future changes in health care systems: In fact, we can actively shape these systems! This helps our patients, because we can provide them with comprehensive care. And in turn, helping our patients is the key to our business success.”
A large shareholder majority of 88.27 percent approved a 10 percent increase in the dividend, from €0.96 to €1.06, the company’s 21st consecutive dividend increase. Shareholder majorities of 99.23 and 95.46 percent, respectively, approved the actions of the Management and Supervisory Boards in 2017.
At the Annual General Meeting, 80.46 percent of the subscribed capital was represented.
Dr. Gerd Krick, Chairman of the Supervisory Board of Fresenius Medical Care AG & Co. KGaA, announced that he will resign from the Supervisory Board, effective at the meeting’s end. He will maintain his position on the Supervisory Board of the General Partner, Fresenius Medical Care Management AG.
Dr. Krick had been Chief Executive Officer of Fresenius Medical Care from 1996, when the company was founded, to 1998, laying the foundation for its global success. In 1998, he resigned from his CEO position and became Chairman of the Supervisory Board of Fresenius Medical Care. The Supervisory Board and Management Board thank Dr. Krick for his tremendous efforts to date and many years of service to the benefit of the company.
The Supervisory Board has appointed Dr. Dieter Schenk, Vice Chairman of the Supervisory Board of Fresenius Medical Care AG & Co. KGaA, as the new Chairman. A new member of the Supervisory Board will be appointed in due course and proposed for election at the next Annual General Meeting, which is expected to take place on May 16, 2019.
Disclaimer
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Fresenius Medical Care AG & Co. KGaA does not undertake any responsibility to update the forward-looking statements in this release.
Q1/2018:
- Sales: €8.1 billion (-1%, +7% in constant currency1)
- EBIT2: €1,054 million (-13%, -5% in constant currency)
- EBIT2: €1,089 million (-10%, -2% in constant currency) (excluding biosimilars business)
- Net income2,3: €450 million (-2%, +7% in constant currency)
- Net income2,3: €476 million (+4%, +12% in constant currency) (excluding biosimilars business)
1 Growth rates adjusted for IFRS 15 adoption (Q1/17 base: €8,223 million)2 Before special items (i.e., expenses related to (i) the Akorn transaction, and (ii) the re-valuation of Sound Physicians’ share-based payment program caused by its announced divestiture)3 Net income attributable to shareholders of Fresenius SE & Co. KGaAFor a detailed overview of special items please see the reconciliation table on page 16 of the PDF.
Stephan Sturm, CEO of Fresenius, said: “We have started into the year with great momentum. All four Fresenius business segments and all regions showed healthy organic growth in the first quarter. Once again, Fresenius Kabi stood out with a strong development across all regions and product areas. From this position of strength, we are well on track to reach our ambitious 2018 growth targets and achieve yet another record year.”
Fresenius terminates merger agreement with Akorn
Fresenius decided on April 22, 2018, to terminate the company’s merger agreement with Akorn, due to Akorn’s failure to fulfill several closing conditions. Fresenius’ decision is based on, among other factors, material breaches of FDA (Food and Drug Administration) data integrity requirements relating to Akorn’s operations found during Fresenius’ independent investigation. Fresenius had offered to delay its decision in order to allow Akorn additional opportunity to complete its own investigation and present any information it wished Fresenius to consider, but Akorn declined that offer.
Akorn disagrees with Fresenius’ position and has filed a corresponding complaint. Fresenius in turn has filed a counterclaim on April 30, 2018.
Stephan Sturm, CEO of Fresenius, said: “We will continue to pursue the strategic goal of expanding our portfolio of injectable generic drugs in North America. This strategy has been, and remains, valid. And given the continuing strong development of Fresenius Kabi in North America, we are in an excellent position to achieve it.”
Group guidance1 for 2018 confirmed
Fresenius confirms its guidance for 2018. Group sales are expected to increase by 5% to 8%2 in constant currency. Net income3,4 is expected to grow by 6% to 9% in constant currency. Excluding expenditures for the further development of the biosimilars business, net income3,5 is expected to grow by ~10% to 13% in constant currency.
Fresenius expects to further reduce its net debt/EBITDA6 ratio by year-end 2018.
7% sales growth in constant currency7
Group sales decreased by 1%7 (increased by 7%7 in constant currency) to €8,121 million (Q1/2017: €8,362 million). Organic sales growth was 4%. Acquisitions/divestitures contributed net 3% to growth. Sales growth was impacted by the anticipated decline in the pharmacy business within Care Coordination at Fresenius Medical Care North America. Also at Fresenius Medical Care, the prior-year quarter saw the compensation for treatments of U.S. war veterans in previous years ("VA agreement"), contributing €100 million as a one-time effect. Negative currency translation effects (8%) were mainly driven by the devaluation of the U.S. dollar and the Chinese yuan against the euro.
1 Excluding effects of the Akorn, NxStage and Sound Physicians transactions2 2017 base adjusted for IFRS 15 adoption (deduction of €486 million at Fresenius Medical Care)3 Net income attributable to shareholders of Fresenius SE & Co. KGaA4 2017 base: €1,816 million; 2018 before special items (i.e., transaction-related effects); including expenditures for further development of biosimilars business (€43 million after tax in FY/17 and ~€120 million after tax in FY/18)5 2017 base: €1,859 million; 2018 before special items (i.e., transaction-related effects)6 Calculated at expected annual average exchange rates, for both net debt and EBITDA; excluding effects of the Akorn, NxStage and Sound Physicians transactions; excluding further potential acquisitions; at current IFRS rules7 Growth rates adjusted for IFRS 15 adoption (Q1/17 base: €8,223 million)
Group sales by region:


7% net income1,2 growth in constant currency
Group EBITDA2 decreased by 10% (-2% in constant currency) to €1,403 million (Q1/2017: €1,560 million). Group EBIT2 decreased by 13% (-5% in constant currency) to €1,054 million (Q1/2017: €1,216 million). The prior-year quarter was strongly influenced by a positive one-time effect: the VA agreement contributed €99 million, or 10%, to EBIT growth in constant currency in Q1/17. The EBIT margin2 was 13.0% (12.7% before IFRS 15; Q1/2017: 14.5%). Group EBIT2 before expenses for the further development of the biosimilars business decreased by 10% (-2% in constant currency) to €1,089 million. Group EBIT2 before VA agreement and excluding the expenses for the biosimilars business increased by 6% in constant currency.
Group net interest2 was -€146 million (Q1/2017: -€157 million). The decrease is mainly driven by currency effects and positive refinancing activities.
The decrease of the Group tax rate before special items to 21.0% (Q1/2017: 29.1%) was mainly due to the U.S. tax reform and a one-time tax effect at Fresenius Medical Care.
Noncontrolling interest2 was €267 million (Q1/2017: €294 million), of which 95% was attributable to the noncontrolling interest in Fresenius Medical Care.
Group net income1,2 decreased by 2% (increased by 7% in constant currency) to €450 million (Q1/2017: €457 million). Earnings per share1,2 decreased by 2% (increased by 6% in constant currency) to €0.81 (Q1/2017: €0.83).
Group net income1,2 before expenses for the further development of the biosimilars business increased by 4% (12% in constant currency) to €476 million (Q1/2017: €457 million). Earnings per share1,2 before expenses for the further development of the biosimilars business increased by 4% (11% in constant currency) to €0.86 (Q1/2017: €0.83).
Group net income1 after special items decreased by 4% (increased by 4% in constant currency) to €440 million (Q1/2017: €457 million). Earnings per share1 after special items decreased by 5% (increased by 4% in constant currency) to €0.79 (Q1/2017: €0.83).
1 Net income attributable to shareholders of Fresenius SE & Co. KGaA2 Before special itemsFor a detailed overview of special items please see the reconciliation table on page 16 of the PDF.
Continued investment in growth
Spending on property, plant and equipment was €380 million (Q1/2017: €328 million), primarily for the modernization and expansion of dialysis clinics, production facilities as well as hospitals and day clinics. This corresponds to 4.7% of sales.
Total acquisition spending was €192 million (Q1/2017: €6,083 million). The prior-year quarter included the acquisition of Quirónsalud.
Cash flow development
Operating cash flow decreased by 50% to €236 million (Q1/2017: €476 million) with a margin of 2.9% (Q1/2017: 5.7%). The decrease is mainly attributable to prior years’ received payment under the VA agreement of ~€200 million as well as to the seasonality in invoicing at Fresenius Medical Care North America, which is not expected to impact full year 2018 cash flow.
Free cash flow before acquisitions and dividends decreased to -€155 million (Q1/2017: €148 million). Free cash flow after acquisitions and dividends was -€389 million (Q1/2017: -€5,393 million).
Solid balance sheet structure
The Group’s total assets increased by 1% (2% in constant currency) to €53,502 million (Dec. 31, 2017: €53,133 million). Current assets grew by 6% (8% in constant currency) to €13,409 million (Dec. 31, 2017: €12,604 million). Non-current assets decreased by 1% (0% in constant currency) to €40,093 million (Dec. 31, 2017: € 40,529 million).
Total shareholders’ equity increased by 1% (3% in constant currency) to €22,020 million (Dec. 31, 2017: €21,720 million). The equity ratio increased to 41.2% (Dec. 31, 2017: 40.9%).
Group debt increased by 1% (2% in constant currency) to €19,200 million (Dec. 31, 2017: € 19,042 million). Group net debt increased by 2% (3% in constant currency) to € 17,716 million (Dec. 31, 2017: € 17,406 million).
As of March 31, 2018, the net debt/EBITDA ratio was 2.98 , (December 31, 2017: 2.841,2).
1 At LTM average exchange rates for both net debt and EBITDA; pro forma closed acquisitions, excluding Akorn, NxStage and Sound Physicians transactions2 Before special itemsFor a detailed overview of special items please see the reconciliation table on page 16 of the PDF.
Increased number of employees
As of March 31, 2018, the number of employees increased by 1% to 275,674 (Dec. 31, 2017: 273,249).
Business Segments
Fresenius Medical Care
Fresenius Medical Care is the world's largest provider of products and services for individuals with renal diseases. As of March 31, 2018, Fresenius Medical Care was treating 322,253 patients in 3,790 dialysis clinics. Along with its core business, the company provides related medical services in the field of Care Coordination.


- Q1/2018 growth impacted by significant currency headwinds and positive one-time effect in prior years’ quarter
- 2018 outlook of net income growth4,7 of 13 to 15% in constant currency confirmed
- 2018 sales growth8 target adjusted to 5 to 7% at constant currency (previously ~8%), mainly due to recent reduction in dosing of calcimimetic drugs in the U.S.
Reported sales were strongly impacted by headwinds from foreign exchange rates and by the anticipated decline in the pharmacy business within Care Coordination at Fresenius Medical Care North America. Sales decreased by 10%1 (increased by 2%1 in constant currency) to €3,976 million (Q1/2017: €4,548 million). Organic sales growth was 3%. Acquisitions/divestitures and the VA agreement in the prior-year quarter decreased sales by 1%. Currency translation effects reduced sales by 12%. Excluding the VA agreement in the prior-year quarter, sales growth1 was 4% in constant currency.
1 Growth rate adjusted for IFRS 15 adoption (Q1/17 base: €4,409 million)2 Excluding VA agreement: 4%3 Adjusted for re-valuation of Sound Physicians’ share-based payment program and excluding VA agreement: 3%4 Net income attributable to shareholders of Fresenius Medical Care AG & Co. KGaA5 Adjusted for re-valuation of Sound Physicians’ share-based payment program, the effect of the U.S. Tax Reform and excluding VA agreement: 8%6 Consistent with guidance, adjusted for re-valuation of Sound Physicians’ share-based payment program, including the effect of the U.S. Tax Reform and including VA agreement7 2017 base: €1,280 million; 2018 including benefits from U.S. tax reform and adjusted for the Sound valuation impact8 2017 reported sales: €17,784 million, adjusted for IFRS 15 adoption (deduction of €486 million)
Health Care services sales (dialysis services and care coordination) decreased by 12%1 (increased by 1%1 in constant currency) to €3,209 million (Q1/2017: €3,769 million). Health Care product sales decreased by 2% (increased by 6% in constant currency) to €767 million (Q1/2017: €779 million).
In North America, sales decreased by 14%1 (-1%1 in constant currency) to €2,774 million (Q1/2017: €3,375 million). Health Care services sales decreased by 14%1 (-1%1 in constant currency) to €2,590 million (Q1/2017: €3,165 million) mainly due to the prior-year VA agreement (€100 million). Excluding the 2017 effect from the VA Agreement Health Care services sales increased by 2%1 in constant currency. Health Care product sales decreased by 12% (increased by 1% in constant currency) to €184 million (Q1/2017: €210 million).
Sales outside North America increased by 2% (10% in constant currency) to €1,198 million (Q1/2017: €1,169 million). Health Care services sales increased by 2% (12% in constant currency) to €619 million (Q1/2017: €604 million). Health Care product sales increased by 3% (8% in constant currency) to €579 million (Q1/2017: €564 million).
Fresenius Medical Care’s EBIT decreased by 24% (-15% in constant currency) to €497 million (Q1/2017: €651 million). The EBIT margin was 12.5% (Q1/2017: 14.3%). Adjusted for the effect of the implementation of IFRS 15, the re-valuation of Sound Physicians’ share-based payment program in connection with the announced divestiture of Sound Physicians and for the positive effect of the VA Agreement in Q1/2017, EBIT was up by 3% in constant currency and EBIT margin was stable at 12.8%.
Net income2 of Fresenius Medical Care decreased by 10% (0% in constant currency) to €279 million (Q1/2017: €308 million). Consistent with guidance, i.e. adjusted for the re-valuation of Sound Physicians’ share-based payment program, net income growth2 was 5% in constant currency. Adjusted for the re-valuation of Sound Physicians’ share-based payment program and the effect of the U.S. Tax Reform in 2018 and for the positive effect of the VA agreement, net income growth2 was 8% in constant currency.
1 Growth rate adjusted for IFRS 15 adoption (Q1/17: deduction of €139 million)2 Net income attributable to shareholders of Fresenius Medical Care AG & Co. KGaA
Operating cash flow was -€45 million (Q1/2017: €170 million). The cash flow margin was 1.1% (Q1/2017: 3.7%). The decrease is mainly attributable to prior years’ payment under the VA agreement of ~€200 million as well as to the seasonality in invoicing at Fresenius Medical Care North America, which is not expected to impact full year 2018 cash flow.
Mainly driven by the change in dosing of calcimimetic drugs, Fresenius Medical Care expects sales to grow by 5 to 7%1 (previously: ~8%1 ) in constant currency. Fresenius Medical Care expects net income2 growth of 13% to 15%3 in constant currency and excluding special items of 7% to 9%4.
The 2018 targets are based on 2017 figures adjusted for the adoption of IFRS 15 implementation and exclude effects from the planned acquisition of NxStage Medical and the announced divestiture of Sound Physicians.
1 2017 reported sales: €17,784 million, adjusted for IFRS 15 adoption (deduction of €486 million)2 Net income attributable to shareholders of Fresenius Medical Care AG & Co. KGaA3 2017 base: €1,280 million; 2018 including benefits from U.S. tax reform and adjusted for the Sound valuation impact4 VA Agreement, Natural Disaster Costs, FCPA related charge, U.S. Tax Reform
For further information, please see Fresenius Medical Care’s Investor News at www.freseniusmedicalcare.com
Fresenius Kabi
Fresenius Kabi offers intravenously administered generic drugs, clinical nutrition and infusion therapies for seriously and chronically ill patients in the hospital and outpatient environments. The company is also a leading supplier of medical devices and transfusion technology products. In the biosimilars business, we are developing products with a focus on oncology and autoimmune diseases.


- Excellent start to 2018
- 9% organic sales growth; 10% EBIT1 growth in constant currency (excluding biosimilars business)
- Strong negative currency translation effects
- Strong operating cash flow
- 2018 outlook confirmed
With €1,603 million (Q1/2017: €1,604 million), sales of Fresenius Kabi were on prior years level (increased by 9% in constant currency). Organic sales growth was 9%. Strong negative currency translation effects (-9%) were mainly related to the devaluation of the U.S. dollar and the Chinese yuan against the euro.
Sales in Europe grew by 2% (organic growth: 3%) to €557 million (Q1/2017: €544 million).
Sales in North America decreased by 5% (organic growth: 10%) to €591 million (Q1/2017: €619 million).
Sales in Asia-Pacific increased by 8% (organic growth: 15%) to €301 million (Q1/2017: €280 million). Sales in Latin America/Africa decreased by 4% (organic growth: 10%) to €154 million (Q1/2017: €161 million).
EBIT1 decreased by 14% (-2% in constant currency) to €268 million (Q1/2017: €313 million). The EBIT margin1 was 16.7% (Q1/2017: 19.5%).
1 Before special items2 Before expenses for the further development of the biosimilars business: 10%3 Net income attributable to shareholders of Fresenius SE & Co. KGaA4 Before expenses for the further development of the biosimilars business: 16%For a detailed overview of special items please see the reconciliation table on page 16 of the PDF.
EBIT1 before expenses for the further development of the biosimilars business decreased by 3% (increased by 10% in constant currency) to €303 million (Q1/2017: €313 million). The EBIT margin1 before expenses for the further development of the biosimilars business was 18.9% (Q1/2017: 19.5%).
Net income1,2 decreased by 11% (increased by 3% in constant currency) to €170 million (Q1/2017: €191 million).
Operating cash flow increased by 18% to €226 million (Q1/2017: €192 million). The cash flow margin was 14.1% (Q1/2017: 12.0%).
Fresenius Kabi confirms its outlook for 2018 and expects organic sales growth of 4% to 7% and EBIT growth in constant currency of -3% to -6%3. Excluding expenditures for the further development of the biosimilars business, EBIT is expected to grow by ~2% to 5%4 in constant currency.
1 Before special items2 Net income attributable to shareholders of Fresenius SE & Co. KGaA3 2017 base: €1,177 million; 2018 before special items (i.e., transaction-related expenses), including expenditures for the further development of the biosimilars business (€60 million in FY/17 and expected expenditures of ~€160 million in FY/18)4 2017 base: €1,237 million; 2018 before special items (i.e., transaction-related expenses)For a detailed overview of special items please see the reconciliation table on page 16 of the PDF.
Fresenius Helios
Fresenius Helios is Europe's leading private hospital operator. The company comprises Helios Germany and Helios Spain (Quirónsalud). Helios Germany operates 111 hospitals, thereof 88 acute care clinics and 23 post-acute care clinics, and treats more than 5.3 million patients annually. Quirónsalud operates 45 hospitals, 55 outpatient centers and around 300 occupational risk prevention centers, and treats approximately 11.6 million patients per year.


- 3% organic sales growth
- 9% EBIT increase
- 2018 outlook confirmed
Fresenius Helios increased sales by 16% to €2,331 million (Q1/2017: €2,018 million). Organic sales growth was 3%. The acquisition of Quirónsalud contributed 13% to sales growth. Helios Spain (Quirónsalud) has been consolidated since February 1, 2017.
Sales of Helios Germany increased by 3% (organic growth: 3%) to €1,574 million (Q1/2017: €1,528 million). Helios Spain increased sales by 54% (organic growth: 1%) to €757 million (Q1/2017: €490 million), mainly due to the additional month of consolidation compared to the prior-year quarter.
Fresenius Helios grew EBIT by 9% to €278 million (Q1/2017: €255 million). The EBIT margin was 11.9% (Q1/2017: 12.6%).
EBIT of Helios Germany decreased by 2% to €177 million (Q1/2017: €181 million) with a margin of 11.2% (Q1/2017: 11.8%). The decline is due to preparatory measures for anticipated regulatory structural requirements for minimum staffing as well as catalogue effects. The anticipated regulatory requirements will be countered by clustering.
EBIT of Helios Spain increased by 39% to €103 million (Q1/2017: €74 million), mainly due to the additional month of consolidation compared to prior-year quarter. The EBIT margin was 13.6% (Q1/2017: 15.1%).
Net income1 of Fresenius Helios increased by 6% to €191 million (Q1/2017: €181 million).
Operating cash flow was €97 million (Q1/2017: €184 million). The margin was 4.2% (Q1/2017: 9.1%).
Fresenius Helios confirms its outlook for 2018 and expects organic sales growth of 3% to 6% and EBIT growth of 7% to 10%.
1 Net income attributable to shareholders of Fresenius SE & Co. KGaA
Fresenius Vamed
Fresenius Vamed manages projects and provides services for hospitals and other health care facilities worldwide. The portfolio ranges along the entire value chain: from project development, planning, and turnkey construction, via maintenance and technical management, to total operational management.


- 9% organic sales growth
- Order backlog of €2,391 million at all-time high
- 2018 outlook confirmed
Sales increased by 12% (12% in constant currency) to €249 million (Q1/2017: €223 million). Organic sales growth was 9%. Sales in the project business increased by 19% to €92 million (Q1/2017: €77 million). Sales in the service business grew by 8% to €157 million (Q1/2017: €146 million).
EBIT of €6 million was unchanged from the prior-year level.
Net income1 of €4 million was also unchanged from prior-year level.
Order intake was €260 million (Q1/2017: €220 million). As of March 31, 2018, order backlog was €2,391 million (December 31, 2017: €2,147 million).
For 2018, Fresenius Vamed expects organic sales growth in the range of 5% to 10% and EBIT growth of 5% to 10%.
1 Net income attributable to shareholders of VAMED AG
Conference Call
As part of the publication of the results for the first quarter 2018, a conference call will be held on May 3, 2018 at 2 p.m. CET (8 a.m. EST). All investors are cordially invited to follow the conference call in a live broadcast over the Internet at www.fresenius.com/investors. Following the call, a replay will be available on our website.
For additional information on the performance indicators used please refer to www.fresenius.com/alternative-performance-measures.
This release contains forward-looking statements that are subject to various risks and uncertainties. Future results could differ materially from those described in these forward-looking statements due to certain factors, e.g. changes in business, economic and competitive conditions, regulatory reforms, results of clinical trials, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. Fresenius does not undertake any responsibility to update the forward-looking statements in this release.
- Revenue and net income growth in the first quarter impacted by significant currency headwind
- Growth delivered by Products business and International segments
- On track to achieve net income growth target
- Expected revenue growth softened due to reduced dosing of calcimimetic drugs in the United States
- Divestiture of Sound Inpatient Physicians to further focus U.S. Care Coordination activities

“On the back of a solid first quarter, which showed healthy organic growth in our businesses, we are heading towards another record year in our company’s history. While managing the shift of the calcimimetic drugs into our clinical operations in the United States, we achieved good organic revenue growth in our Dialysis Services business and strong organic revenue growth in our Products business. This provides a solid basis to deliver on our growth targets for this year. With the planned sale of Sound Inpatient Physicians we have narrowed the focus of our Care Coordination strategy in the U.S. on areas that provide the highest contribution and the best outcomes for our patients,” said Rice Powell, Chief Executive Officer of Fresenius Medical Care. “I am proud to announce that we have just proven once again our commitment to patients. We have received the highest quality rankings in the industry from the U.S. Centers for Medicare and Medicaid Services last week.”
Currency headwind impacts revenue and earnings, healthy organic growth continues
Revenue in the first quarter 2018 was significantly impacted by a 12% negative impact resulting from foreign currency translation, declining 1% at constant currency to EUR 3,976 million. Adjusting Q1 2017 for the prior year impact from the recognition of revenue related to the agreement with the U.S. Departments of Veterans Affairs and Justice (VA Agreement) and for the IFRS 15 implementation, revenue growth in the first quarter 2018 was 4% at constant currency. Health Care Services revenue declined by 3% at constant currency (EUR 3,209 million), driven by the effect of the implementation of IFRS 15. Health Care Products revenue increased by 6% at constant currency to EUR 767 million. Organic growth for Health Care Services was at 2%, and for the Health Care Products business at 6%. Dialysis treatments increased by 3% as a result of growth in same-market treatments (2%) and contributions from acquisitions (1%).
Total operating income (EBIT) reached EUR 497 million (margin of 12.5%, 180 basis points below the level of last year). This development was strongly impacted by the VA Agreement and the impact from the initial increase in valuation of Sound Physicians’ share based payment program caused by the sale of Sound Physician (initial Sound valuation impact). Adjusting for the revenue impact from the implementation of IFRS 15 and excluding the VA Agreement as well as the initial Sound valuation impact, EBIT grew by 3% at constant currency and EBIT margin was stable at 12.8%.
Net income attributable to shareholders of Fresenius Medical Care AG & Co. KGaA remained strong with EUR 279 million, a stable development at constant currency. Adjusted for the negative impact of the initial Sound valuation impact net income grew by 5% on a constant currency basis (EUR 292 million). Excluding all special items – namely the VA Agreement in Q1 2017 and the positive effect from the U.S. Tax Reform in Q1 2018 as well as the initial Sound valuation impact, net income attributable to shareholders of Fresenius Medical Care AG & Co. KGaA increased by 8% at constant currency.
Based on the number of approximately 306.5 million shares (weighted average number of shares outstanding), basic earnings per share (EPS) amounted to EUR 0.91, compared to EUR 1.01 for the first quarter of 2017.
Development of Reporting Segments
North America revenue, which corresponds to 70% of total revenue, was down by 5% at constant currency to EUR 2,774 million.
Dialysis Care revenue decreased by 16% to EUR 2,075 million, including a 13% negative impact resulting from foreign currency translation. At constant currency, Dialysis Care revenue decreased by 3%, mainly due to the implementation of IFRS 15 (EUR 88 million) and the prior-year impact from the VA Agreement (EUR 100 million). Excluding the 2017 effect from the VA Agreement and the 2017 effect from the implementation of IFRS 15, Dialysis Care revenue increased by 5% at constant currency. Same-market treatments grew by 2%, organic revenue per treatment by 2% and acquisitions contributed 1%. At constant currency, Care Coordination revenue decreased by 14%, driven by the shift of calcimimetic drugs into the clinical environment, the implementation of IFRS 15 and the impact from the Shiel Laboratories divestiture in Q4 2017.
As of the end of March 2018, the company was treating 197,339 patients at its 2,419 clinics in North America. Both numbers increased by 4%, while dialysis treatments increased by 3%.


In the U.S., the average revenue per treatment, adjusted for the implementation of IFRS 15 and excluding the 2017 impact of the VA Agreement, the average revenue per treatment increased by USD 6 from USD 342 to USD 348. The increase was mainly driven by the announced initial introduction of calcimimetic drugs in the clinical environment, which is still in an early conversion stage. Higher implicit price concessions (IFRS 15) and, as previously indicated, lower revenue from commercial payors mitigated this effect.
Cost per treatment in the U.S., adjusted for the implementation of IFRS 15, increased to USD 288 (from USD 276). This development was largely a result of the announced initial introduction of calcimimetic drugs in the clinical environment, which is still in an early conversion stage, higher personnel expense, as well as increased property and other occupancy related costs and increased costs for medical supplies partially offset by lower costs for health care supplies.
At constant currency, Health Care Products revenue increased by 1% due to higher sales of renal pharmaceuticals, peritoneal dialysis products, hemodialysis solutions and concentrates, partially offset by lower sales of machines and dialyzers.
The total operating income of the North America segment was EUR 362 million (-31%), (-21% at constant currency), an operating income margin of 13.1%. Adjusted for the initial Sound valuation impact and the 2017 effects from the VA Agreement, operating income (EBIT) was EUR 375 million compared to EUR 427 million in the first quarter 2017. The respective operating income margin was 13.5% compared to 13.6% in the first quarter of 2017.
EMEA revenue increased by 6% at constant currency to EUR 636 million, mainly driven by positive business development in Health Care Services revenue and Health Care Products revenue, which both increased by 6% at constant currency. The increase in Health Care Services revenue was driven by acquisitions, same-market treatment growth and an increase in dialysis days. Dialysis Products revenue grew by 7% at constant currency to EUR 302 million, due to higher sales of products for acute care treatments, machines, peritoneal dialysis products and renal pharmaceuticals.
Non-dialysis Products revenue decreased by 6% to EUR 20 million, primarily due to lower sales of acute cardiopulmonary products. There was virtually no impact from foreign currency translation effects. Operating income was EUR 109 million. The operating income margin decreased from 18.7% to 17.1%, mainly due to unfavorable impacts from currency effects partially offset by the impact of one additional dialysis day.
As of the end of March 2018, the company had 63,114 patients (5% increase) being treated at 754 clinics (4% increase) in the EMEA region. Dialysis treatments increased by 5%.
Asia-Pacific revenue grew by 14% at constant currency to EUR 392 million. In the region, Health Care Services revenue increased 9% (20% at constant currency) to EUR 184 million. Care Coordination activities contributed EUR 46 million to Health Care Services revenue. With growth of 8% in constant currency to EUR 208 million, the Health Care Products business showed a solid sales performance, mainly driven by higher sales of chronic hemodialysis products and products for acute care treatments. Operating income reached EUR 74 million (Q1 2017: EUR 82 million). The operating income margin decreased to 19.0% in Q1 2018 (Q1 2017: 21.7%). This was primarily driven by foreign currency transaction effects and delayed product sales.
As of the end of March 2018, the company had 30,194 patients (2% increase) being treated at 385 clinics in Asia-Pacific. Dialysis treatments increased by 2%, impacted by same market treatment growth of 4% partially offset by the effect of sold or closed clinics.
Latin America delivered revenue of EUR 170 million, a significant improvement of 17% at constant currency. This growth was mainly driven by an increase in organic revenue per treatment. Health Care Products revenue grew by 25% at constant currency based on higher sales of dialyzers, machines, products for acute treatments and peritoneal dialysis products. With an operating income of EUR 14 million the segment generated an operating income on previous year’s level. Operating income margin increased slightly to 8.3% in Q1 2018 (Q1 2017: 8.1%).
As of the end of March 2018, the company was treating 31,606 patients (5% increase) at 232 clinics in Latin America. Dialysis treatments increased by 4%.
Net interest expense was EUR 80 million compared to EUR 92 million in the first quarter of 2017, a decrease of 14% (5% at constant currency). The decrease was driven by the lower leverage level and a repayment of high interest-bearing senior notes. Income tax expense was EUR 87 million for the first quarter of 2018, which translates into an effective tax rate of 20.9%, compared to last year’s Q1 with a tax rate of 32.5%. The strong reduction was largely driven by the U.S. Tax Reform.
Cash flow with normalized development
In the first quarter of 2018, the company used EUR 45 million in net cash from operating activities, compared to EUR 170 million provided by operating activities in last year’s Q1. The decrease in net cash provided by operating activities was largely driven by the positive impact from the 2017 cash inflow related to the VA Agreement, a higher impact from seasonality in invoicing and increased inventory levels, partially offset by a positive impact from lower income tax payments. The number of days sales outstanding (DSOs) increased sequentially by 10 days compared with Q4 2017 to reach 85 days driven by the above mentioned seasonality in invoicing. Free cash flow (Net cash used in operating activities, after capital expenditures, before acquisitions and investments) amounted to EUR (263 million) and EUR (25 million) for the three months ended March 31, 2018 and March 31, 2017, respectively. Free cash flow in percent of revenue was (6.6%) and (0.6%) for the three months ended 2018 and 2017, respectively.
Focusing the profile in U.S. Care Coordination portfolio
On April 21, Fresenius Medical Care announced to divest its controlling interest in Sound Inpatient Physicians to an investment consortium for total transaction proceeds of USD 2.15 billion (EUR 1.76 billion3). The divestment is expected to generate a pre-tax book gain of approximately EUR 800 million3,4. Closing of the transaction is subject to regulatory approvals and anticipated late in 2018.
Employees
As of March 31, 2018, Fresenius Medical Care had 114,831 employees (full-time equivalents) worldwide, compared to 110,530 employees at the end of March 2017. This increase was mainly attributable to our continued organic growth and acquisitions.
Outlook 2018
The company expects revenue5 growth between 5% and 7% at constant currency. Adjusted net income6 is expected to increase by 13% to 15% at constant currency and excluding special items7 to increase by 7% to 9%.
The targets exclude effects from major transactions such as the planned acquisition of NxStage Medical and the planned divestiture of Sound Physicians.
Conference call
Fresenius Medical Care will host a conference call to discuss the results of the first quarter today at 3:30 p.m. CEDT / 9:30 a.m. EDT. Details will be available on the company’s website www.freseniusmedicalcare.com in the “Investors/Events” section. A replay will be available shortly after the call.
1For a detailed reconciliation, please refer to the table at the end of the press release
2Net income attributable to shareholders of Fresenius Medical Care AG & Co. KGaA
3EUR/USD 1.22
4Based on the company’s latest available valuation of the North American operating segment
52017 adjusted for the effect of IFRS 15 implementation
6Attributable to shareholders of Fresenius Medical Care AG & Co. KGaA, adjusted for the Sound valuation impact
7VA Agreement, Natural Disaster Costs, FCPA related charge, U.S. Tax Reform
Disclaimer
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Fresenius Medical Care AG & Co. KGaA does not undertake any responsibility to update the forward-looking statements in this release.
Fresenius Medical Care, the world’s largest provider of dialysis products and services, today announced a leadership change in its EMEA (Europe, Middle East and Africa) region. Effective 1 September 2018, Ms. Katarzyna Mazur-Hofsäss, Ph.D., will assume the Management Board position in charge of EMEA. She follows Dominik Wehner, who decided to step down from his position for personal reasons, effective on 31 December 2017. In the interim period Rice Powell, Chief Executive Officer of Fresenius Medical Care and Chairman of the Management Board, manages the EMEA region.
“I want to thank Dominik for his more than 20 years of contributions – most notably his commitment of caring for our patients at all times while enhancing our geographical footprint in the EMEA region. We are very proud of what Dominik has achieved and wish him all the best for his future,” said Rice Powell.
Katarzyna Mazur-Hofsäss has been president for EMEA at the med-tech company Zimmer Biomet since 2013. In her 25 years of professional career she has gained extensive experience in the medical and pharma industry from her positions at Abbott Laboratories and Roche. Katarzyna Mazur-Hofsäss is a physician by educational background and holds a Ph.D. from Gdansk Medical University in Poland, as well as an MBA from the Warsaw School of Economics and the University of Minnesota. “Katarzyna will not only be well equipped to run our expanding EMEA region but she will also have a fresh perspective that can provide new impulses. We look forward to welcoming her in our team,” said Rice Powell.
Disclaimer
This release contains forward-looking statements that are subject to various risks and uncertainties. Actual results could differ materially from those described in these forward-looking statements due to certain factors, including changes in business, economic and competitive conditions, regulatory reforms, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. These and other risks and uncertainties are detailed in Fresenius Medical Care AG & Co. KGaA's reports filed with the U.S. Securities and Exchange Commission. Fresenius Medical Care AG & Co. KGaA does not undertake any responsibility to update the forward-looking statements in this release.
Fresenius has decided today to terminate the company’s merger agreement with Akorn, due to Akorn’s failure to fulfill several closing conditions.
Fresenius’ decision is based on, among other factors, material breaches of FDA1 data integrity requirements relating to Akorn’s operations found during Fresenius’ independent investigation. Fresenius offered to delay its decision in order to allow Akorn additional opportunity to complete its own investigation and present any information it wished Fresenius to consider, but Akorn has declined that offer.
Fresenius confirms its guidance for 2018. The Group continues to expect a sales growth of 5% to 8%2 in constant currency. Group net income3 is expected to increase by 6% to 9%4 in constant currency (excluding expenditures for the further development of the biosimilars business around 10% to 13%5).
On Saturday, Fresenius Medical Care announced the sale of Sound Inpatient Physicians Holdings, LLC. The expected pre-tax book gain of around €800 million on this transaction is excluded from Fresenius’ 2018 Group guidance.
Fresenius will report its first-quarter results, as scheduled, on May 3, 2018.
1FDA: Food and Drug Administration
22017 adjusted for IFRS 15 (EUR486 million at Fresenius Medical Care)
3Net income attributable to shareholders of Fresenius SE & Co. KGaA
4Base 2017: EUR1,816 million; 2018 before special items (acquisition-related expenses); including expenditures for further development of biosimilars business (EUR43 million after tax in FY/17 and ~EUR120 million after tax in FY/18)
5Base 2017: EUR1,859 million; 2018 before special items (acquisition-related expenses); excluding expenditures for further development of biosimilars business (EUR43 million after tax in FY/17 and ~EUR120 million after tax in FY/18)
This release contains forward-looking statements that are subject to various risks and uncertainties. Future results could differ materially from those described in these forward-looking statements due to certain factors, e.g. changes in business, economic and competitive conditions, regulatory reforms, results of clinical trials, foreign exchange rate fluctuations, uncertainties in litigation or investigative proceedings, and the availability of financing. Fresenius does not undertake any responsibility to update the forward-looking statements in this release.
Pagination
- Previous page
- Page 26
- Next page










